Chemistry - Online Test
Q1. Acetylation of glucose yields
Answer : Option D
Explaination / Solution:
Acetyl group adds in glucose, giving acetylation reaction.
Q2. For the first order reaction, half life is equal to
Answer : Option C
Explaination / Solution:
For 1st order reaction half life is independent of initial concentration of reactant.
Q3. Hardening of leather in tanning industry is based on
Answer : Option A
Explaination / Solution:
Fact
Q4. Charles’ law states that pressure remaining constant, the volume of a fixed mass of a gas is
Answer : Option B
Explaination / Solution:
P α T (at constant V).
Q5. The dynamic nature of chemical equilibrium can be demonstrated in the synthesis of ammonia by Haber.s process. Choose the appropriate option given below
Answer : Option A
Explaination / Solution:
Use of isotope (deuterium) in the formation of ammonia clearly indicates that chemical reactions reach a state of dynamic equilibrium in which the rates of forward and reverse reactions are equal and there is no net change in composition.
Q6. Ketones react with Grignard reagent to produce
Answer : Option B
Explaination / Solution:
The Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or aldehyde, to form a tertiary or secondary alcohol, respectively. The reaction with formaldehyde leads to a primary alcohol.
Grignard Reagents are also used in the following important reactions: The addition of an excess of a Grignard reagent to an ester or lactone gives a tertiary alcohol in which two alkyl groups are the same, and the addition of a Grignard reagent to a nitrile produces an unsymmetrical ketone via a metalloimine intermediate.
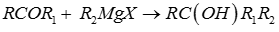
Q7. The main product formed by treating an alkyl or benzyl halide with excess ammonia
Answer : Option B
Explaination / Solution:
The major product is primary amine.
Q8. General formula for alkenes is
Answer : Option C
Explaination / Solution:
Alkenes are unsaturated hydrocarbons with atleast on double bond between the carbon atoms.Their general formula is CnH2n.
Q9. Cryoscopic constant of a liquid is
Answer : Option C
Explaination / Solution:
ΔTf= Kfm. When m(molality )= 1m then kf= ΔTf
Q10. The highest value of oxidation number changes from 1 to 7
Answer : Option D
Explaination / Solution:
The highest value of oxidation number exhibited by an atom of an element generally increases across the period in the periodic table.
