Chemistry - Online Test
Q1. In the reaction, R – X + NaoR’ → ROR’ + X ( – ve ion) The main product formed is
Answer : Option A
Explaination / Solution:
This is normal substitution reaction.
Q2. Oxime is formed by treating glucose with
Answer : Option C
Explaination / Solution:
Oxime is combination of carbonyl group and and hydroxyl groups.
Q3.
For a reaction; pA +qB products, the rate law expression is
Answer : Option B
Explaination / Solution:
Order of a reaction is experimentally determined quantity. It may or may not be equal to stoichiometric coefficients.
Q4. The emulsifying agent present in milk that makes it stable is
Answer : Option B
Explaination / Solution:
fact
Q5. At constant temperature, the pressure of a fixed amount (i.e., number of moles n) of gas is?
Answer : Option A
Explaination / Solution:
From ideal gas equation PV=nRT,
keeping n and T fixed,
P α 1/V. (Boyles Law)
Here, T = Temperature
P = Pressure
α. = sign of Proportionality
V = Volume
n= number of moles of gas
R= Universal gas constant
Q6. When the soda bottle is opened, some of the dissolved carbon dioxide gas escapes because of
Answer : Option D
Explaination / Solution:
Soda water, like other carbonated beverages, contains carbon dioxide that has dissolved under pressure. When the pressure is released by opening the soda container, the liquid cannot hold as much carbon dioxide, so the excess bubbles out of the solution. If the soda is left open, additional carbon dioxide will slowly escape into the air. Under warm conditions, the carbon dioxide leaves the solution faster.
Q7. Aldehydes react with Grignard reagent to produce
Answer : Option A
Explaination / Solution:
The Grignard Reaction is the addition of an organomagnesium halide (Grignard reagent) to a ketone or aldehyde, to form a tertiary or secondary alcohol, respectively. The reaction with formaldehyde leads to a primary alcohol.
Grignard Reagents are also used in the following important reactions: The addition of an excess of a Grignard reagent to an ester or lactone gives a tertiary alcohol in which two alkyl groups are the same, and the addition of a Grignard reagent to a nitrile produces an unsymmetrical ketone via a metalloimine intermediate.
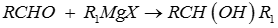
Q8. Quaternary ammonium salt is formed
Answer : Option A
Explaination / Solution:
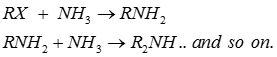
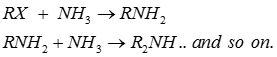
Q9. Halogenation of alkanes is a
Answer : Option A
Explaination / Solution:
Halogenation of alkanes is a substitution reaction because the hydrogen atoms are replaced by halogens in the presence of sunlight to give the corresponding alkyl halides.The reaction follows free radical mechanism.
Q10. Which of the following is a colligative property?
Answer : Option D
Explaination / Solution:
Colligative property is dependent on no. of moles of solute. (Л=cRT)
