Chemistry - Online Test
Q1. Which is the most stable oxidation state of iron?
Q2. Ozonolysis of alkenes followed by reaction with zinc dust and water gives depending on the substitution pattern of the alkene
Answer : Option D
Explaination / Solution:
Reductive ozonolysis yields aldehyde or ketone.
Q3. For the synthesis of methanol and a number of hydrocarbons , the compounds used are
Answer : Option B
Explaination / Solution:
A mixture of CO and H2 in any ratio is called synthesis gas or syn gas as it is used in synthesis of methanol and a number of hydrocarbon.
Q4. Saponification is a process which involves
Answer : Option C
Explaination / Solution:
Saponification is process which involves the preparation of soaps.
Q5. Which of the following complex will give white precipitate with barium chloride solution?
Answer : Option D
Explaination / Solution:
BaSO4 is precipitated when SO42− ion is present outside the coordination sphere.
Q6. The reversible work done by a galvanic cell is equal to decrease in its Gibbs energy and therefore, if the emf of the cell is E and nF is the amount of charge passed and ΔrG is the Gibbs energy of the reaction, then
Answer : Option B
Explaination / Solution:
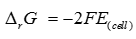
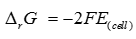
Q7. A qualitative measure of the stability of an ionic compound is provided by
Answer : Option C
Explaination / Solution:
Lattice energy is an estimate of the bond strength in ionic compounds. It is defined as the heat of formation for ions of opposite charge in the gas phase to combine into an ionic solid.
The stability of ionic bond is directly propotional to lattice energy.
Q8. Monovalent sodium and potassium ions found in large proportions in biological fluids. They help in
Answer : Option A
Explaination / Solution:
Na and K ions help in transmission of nerve impulses , in regulating the flow of water accross the cell membranes and in the transport of sugar and amino acids in to cells. so monovalent sodium and potassium ions found in large proportions in biological fluids
Q9. Delocalisation of σ-electrons of C -H bond of an alkyl group directly attached to an atom of unsaturated system or to an atom with an unshared p-orbital is called :
Answer : Option D
Explaination / Solution:
The interaction of the electrons in a sigma bond (usually C–H or C–C) with an adjacent empty (or partially filled) non-bonding p-orbital, antibonding σ or π orbital,or filled π orbital, to give an extended molecular orbital that increases the stability of the system is Hyperconjugation.
Q10. Precision and Accuracy refer to _______ of various measurements for the same quantity repeatedly and agreement of a particular value to the ------- -------- of the result , respectively.
Answer : Option A
Explaination / Solution:
Precision means closeness of all the data values measured while accuracy means closeness of particular value with the real value.
For example ,
when the reported values are 2.01, and 1.99 g as the result and its truevalue is 2.00 g .
These values are both precise and accurate.
