Chemistry - Online Test
Q1. The kelvin scale is related to celsius scale as :
Answer : Option A
Explaination / Solution:
The relation between Kelvin scale & Celcius scale of temperatures is -
K = °C + 273.15
Such a relation is based upon the experimental findings and subsequent conclusion of Charle's law .
The Kelvin scale is also termed as " absolute scale of temperature."
It is interesting to note that temperature below 0o C ( ie. negative values ) are possible in Celcius scale but in Kelvin scale , negative temperature is not possible .
Q2. Bromomethane, Chloromethane, Dibromomethane. 1 – Chloropropane, Isopropyl chloride, 1 – Chlorobutaneare all
Answer : Option C
Explaination / Solution:
These all are covalent compounds hence are soluble in organic solvents.
Q3. The polymer(s) of dextrose is
Answer : Option C
Explaination / Solution:
Cellulose and Stach is polymer of dextrose.
Q4. Which of the following statement is true for order of a reaction?
Answer : Option A
Explaination / Solution:
Order of a reaction is experimentally determined quantity so it can be negative fractional or zero.
Q5. Artificial smoke screens used in the warfare are colloidal dispersion of
Answer : Option A
Explaination / Solution:
Artificial smoke screens used in the warfare are colloidal dispersion of Titanium oxide.
Q6. Gases have much lower density than the solids and liquids because ?
Answer : Option D
Explaination / Solution:
In gases, molecules are far apart from each other, thermal energy which is a measure of freeness of motion is thus very high resulting in a very large volume for the same mass(compared to solid and liquid). Hence there density is lowest.
Q7. The mass of a gas dissolved in a given mass of a solvent at any temperature is proportional to the pressure of the gas above the solvent.
Answer : Option A
Explaination / Solution:
This equilibrium is governed by Henry’s law, which states that the mass of a gas dissolved in a given mass of a solvent at any temperature is proportional to the pressure of the gas above the solvent. This amount decreases with increase of temperature.
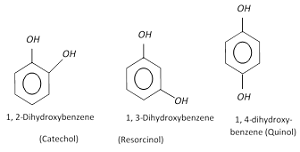
Q8. One of the following is not a dihydroxy derivative of benzene.
Answer : Option A
Explaination / Solution:
- Catechol is o- hydroxyl phenol or Catechol also known as pyrocatechol or 1,2- dihydroxybenzene,
- Resorcinol is m- hydroxyphenol
- Quinol is p-hydroxyphenol or benzene-1,4-diol is an aromatic organic compound that is a type of phenol, a derivative of benzene.
Q9. For producing amines, the reaction of nitro compounds with iron scrap is preferred because
Answer : Option C
Explaination / Solution:
This reaction is preferred because on reduction of nitro compouns to amine , Fe will get oxidize to Fe(II) a which on hydrolysis will produce HCl which is required in the reaction.
Q10. boiling point of alkanes
Answer : Option B
Explaination / Solution:
As molecular mass increases,the magnitude of van der Waals forces of attraction increases and hence boiling point increases accordingly.
