Chemistry - Online Test
Q1. Tropospheric pollution is caused by :
Answer : Option C
Explaination / Solution:
Various oxides of sulphur, nitrogen and carbon like SO2, SO3, NO, NO2, CO and CO2 cause tropospheric pollutionand inflict harm to living beings.
Q2. Transition metals with highest melting point is
Answer : Option A
Explaination / Solution:
W belongs to 5d series and also it have lot of unpaired electrons thus it forms strong metallic bonding.
Q3. Prop – 2 – enal is called
Answer : Option C
Explaination / Solution:
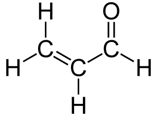
this is Acrolein.

this is Acrolein.
Q4. Heavy hydrogen is also called
Answer : Option A
Explaination / Solution:
Deuterium, (D, or 2H), also called heavy hydrogen, isotope of hydrogen with a nucleus consisting of one proton and one neutron, which is double the mass of the nucleus of ordinary hydrogen (one proton). Deuterium has an atomic weight of 2.014.
Q5. The coordination number of the central ion may be obtained from
Answer : Option D
Explaination / Solution:
Coordination number of the central ion is equal to no. of coordination bonds formed with surrounding ions.
Q6. Ortho – sulphobenzimideis
Answer : Option B
Explaination / Solution:
It is artificial sweetner.
Q7. Relationship between equilibrium constant of the reaction and standard potential of the cell in which that reaction takes place is
Answer : Option B
Explaination / Solution:
At equilibrium E cell becomes zero .
Q8. 788 kJ of energy is required to separate one mole of solid NaCl into one mole of (g) and one mole of (g) to an infinite distance. This process explains
Answer : Option C
Explaination / Solution:
Lattice enthalpy is simply the change in Enthalpy associated with the formation of one mole of an ionic compound from its oppositely charged ions in their standard states under standard conditions.
Q9. The diagonal relationship exists between
Answer : Option A
Explaination / Solution:
Lithium shows diagonal relationship to Magnisium due to their nearly same polarising power, comparable electronegativities(Li=1.00, Mg=1.20) and small difference in radius (Li=152Pm, Mg=159pm).
Q10. Which of the following compounds contain all the carbon atoms in the same hybridisation state?
Answer : Option B
Explaination / Solution:
In option:A,all the 4 carbon atoms are sp-hybridized.
