Chemistry - Online Test
Q1. In general, in the molecules containing multiple bonds,_____ provide the most reactive centres. Choose the most appropriate one.
Answer : Option A
Explaination / Solution:
pie bonds are the most reactive centers.
Q2. SI units for Base Physical Quantities of length, mass and current are
Answer : Option D
Explaination / Solution:
The International System of units ( abbreviated as SI ) is modern form of metric system , and is most widely used form of measurements .
It comprises of a coherent system of measurement built of seven base units .
The base units for length , mass and current in this system of measurements are , " meter , kilogram , ampere " ,symbolised respectively as as
m ,kg , A
Q3. AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as main product
Answer : Option B
Explaination / Solution:
AgCN is covalent in nature while KCN is ionic so KCN produces CN- in the solution.
CN- is an ambient nucleophile i.e. it can form bond from both C and N atoms. KCN is an ionic compound so both C and N sides are available for the formation of bond, but C-C bond is stronger than C-N bond so cynide is formed. AgCN is a covalent compound so only N side is available for CN to form bond and thus isocynide is formed
Q4. The most abundant organic compound on earth
Answer : Option A
Explaination / Solution:
Glucose is most abundant organic compound on earth.
Q5. For which of the following reaction the units of rate constant and rate of the reaction are same?
Answer : Option A
Explaination / Solution:
Rate=k[Reactant]0
Q6. Gelatine is often used as an ingredient in the manufacture of ice cream for
Answer : Option B
Explaination / Solution:
Gelatin is used as ingredient in manufacture of ice cream.
Q7. Three states of matter are the result of?
Answer : Option D
Explaination / Solution:
Three states of matter are the result of balance between intermolecular forces and the thermal energy of the molecules. These two factors differentiate them from each other. For understanding, The strongest intermolecular forces are in solid then in liquid and weakest in gases.
Q8. The boiling point of water at atmospheric pressure
Answer : Option C
Explaination / Solution:
Water and water vapour are in equilibrium position at atmospheric pressure (1.013 bar) and at 100°C in a closed vessel. The boiling point of water is 100°C at 1.013 bar pressure. For any pure liquid at one atmospheric pressure (1.013 bar), the temperature at which the liquid and vapours are at equilibrium is called normal boiling point of the liquid. Boiling point of the liquid depends on the atmospheric pressure.
Q9. Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds.
Answer : Option D
Explaination / Solution:
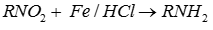
Scrap iron with HCl acts as a reducing agent to reduce nitro group to amine.
Q10. 3 – methyl phenol is called
Answer : Option A
Explaination / Solution:
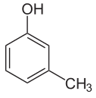
Meta-Cresol (m-cresol) also 3-methylphenol, is an organic compound with the formula
