Chemistry - Online Test
Q1. Correct expression for mass number A is
Answer : Option D
Explaination / Solution:
Mass number. The mass number (symbol A), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus.
mass No. = no. of protons + no. of neutrons.
Q2. Troposphere is a region of the atmosphere which contains :
Answer : Option D
Explaination / Solution:
Troposphere is the lowest layer of the atmosphere where all living beings including humans live. It also has air, much water vapours, clouds and is turbulent in nature.
Q3. Which among the following transition metal has lowest melting point?
Answer : Option B
Explaination / Solution:
Zn has lowest melting point because it doesnot have any unpaired electrons.
Q4. The carbonyl carbon atom
Answer : Option C
Explaination / Solution:
R1R2C=O is carbonyl so 3 sigma bond and 1 pie bond.
Q5. In the earth’s atmosphere, hydrogen exists in the form of
Answer : Option D
Explaination / Solution:
In the combined form it constitutes 15.4% of the earth crust and the ocean.
Q6. Which of the following is most likely structure of CrCl3.6H2O if 1/3 of total chlorine of the compound is precipitated by adding AgNO3 to its aqueous solution?
Answer : Option C
Explaination / Solution:
1 mol of Cl is precipitated so 1 Cl will be present outside the coordination sphere.
Q7. Very low concentrations of Sulphur – dioxide acts as
Answer : Option A
Explaination / Solution:
Very low concentration of act as only disinfectant.
Q8. For a general electrochemical reaction of the type :
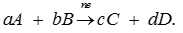
Nernst equation can be written as:
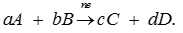
Nernst equation can be written as:
Answer : Option D
Explaination / Solution:
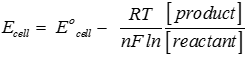
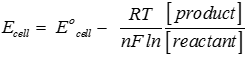
Q9. When a gas phase atom in its ground state gains an electron. This is called
Answer : Option B
Explaination / Solution:
Electron gain enthalpy (ΔegH) is defined as the enthalpy change associated with an isolated gaseous atom (X) when it gains an electron to form its corresponding anion. The reaction can be given as below:
On the basis of nature of the element, the process of accepting electron in an atom can either be exothermic or endothermic.
Q10. One of the following is highly radioactive
Answer : Option A
Explaination / Solution:
Fr is highly radioactive metal, that decays in to astatine,,radium and radon , it is also highly unstable element found only in trace amount on earth.
