Chemistry - Online Test
Q1. Dipole-dipole forces act between the molecules possessing permanent dipole. Ends of dipoles possess ‘partial charges’. The partial charge is
Answer : Option A
Explaination / Solution:
It is less than unit electronic charge.
Q2. Derived protein is.
Answer : Option B
Explaination / Solution:
Peptones are derived protein.
Q3. Which of the following reactions will yield phenol?
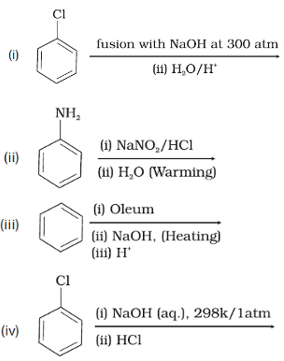

Answer : Option C
Explaination / Solution:
Preparation of phenols from haloarenes: Chlorobenzene is an example of haloarenes which is formed by mono substitution of benzene ring. When chlorobenzene is fused with sodium hydroxide at 623K and 320 atm sodium phenoxide is produced. Finally, sodium phenoxide on acidification gives phenols.
Preparation of phenols from diazonium salts: When an aromatic primary amine is treated with nitrous (NaNO2 + HCl) acid at 273 – 278 K, diazonium salts are obtained. These diazonium salts are highly reactive in nature. Upon warming with water, these diazonium salts finally hydrolyse to phenols. Phenols can also be obtained from diazonium salts by treating it with dilute acids.
Preparation of phenols from benzene sulphonic acid: Benzenesulphonic acid can be obtained from benzene by reacting it with oleum. Benzenesulphonic acid thus formed is treated with molten sodium hydroxide at high temperature which leads to the formation of sodium phenoxide. Finally, sodium phenoxide on acidification gives phenols.
Q4. To neutralize completely 40 mL of 0.1M aqueous solution of KOH, the volume of 0.1M aqueous solution of phosphoric acid required is
Answer : Option A
Explaination / Solution:
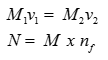
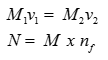
Q5. Calculate the number of unit cells present in 1 g of gold which has face centered cubic lattice.
Answer : Option D
Explaination / Solution:
Number of unit cells =
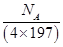
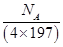
Q6. Hydrogen gas is not liberated when the following metal is added to dil. HCl.
Answer : Option C
Explaination / Solution:
Ag is less reactive than Hydrogen.
Q7. When methylamine reacts with HCl the product is
Answer : Option B
Explaination / Solution:
reacts with HCl to form salt.
Q8. One mole is the amount of a substance that contains as many particles or entities as there are atoms in
Answer : Option C
Explaination / Solution:
One mole is the amount of a substance that contains as many particles or entities as there are atoms in exactly 12 g of the isotope .
A mole of a substance always contains the same number of entities , no matter what the substance may be. In order to determine this number precisely , the mass of carbon -12 atom was determined by mass spectrometer and found to be equal to 1.992648 x 10-23 g. Knowing that one mole of carbon weighs 12 g . the number of atoms in it is equal to :
6.0221367 x 1023
Q9. The general formula of a cycloalkane is
Answer : Option B
Explaination / Solution:
Cycloalkanes have general formula of
Q10. The reducing agent used in the blast furnace to reduce haematite to iron is
Answer : Option A
Explaination / Solution:
CO is used as reducing agent.
