Chemistry - Online Test
Q1. Anti – histamines stop allergic reactions by
Answer : Option D
Explaination / Solution:
Anti – histamines stop allergic reactions by competing with histamine for binding sites of histamine receptor.
Q2. Which of the following reagents can be used to convert a carboxylic acid directly into its corresponding acid chloride derivative?
Answer : Option B
Explaination / Solution:
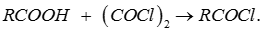
Q3. Li occupies higher position in the electrochemical series of metals as compared to Cu since
Answer : Option A
Explaination / Solution:
Li is alkali metal /Li = -3.045V.``
Q4. The enthalpies of elements in their standard states are taken as zero. The enthalpy of formation of a compound
Answer : Option C
Explaination / Solution:
Enthapy of formation of a compound from elements in standard state can b +ve or –ve.
Q5. In which of the following substances will hydrogen bond be strongest?
Answer : Option A
Explaination / Solution:
Out of all H2O will form strongest hydrogen bonding due to high electronegativity of O.
Q6.
IUPAC name of (CH3)3CCl
Answer : Option B
Explaination / Solution:
Longest chain will be of three carbon to which Cl and CH3 will be attached at 2 position.
Q7. Metal carbonates decompose on heating to give metal oxide and carbondioxide. Which of the metal carbonates is most stable thermally?
Answer : Option D
Explaination / Solution:
is very stable due to size compatibility factor . A larger cation can stablise a larger anion.Group 2 element become more thermally stable down the group.
Q8. In which of the following representations given below spatial arrangement of group/ atom different from that given in structure ‘A’?
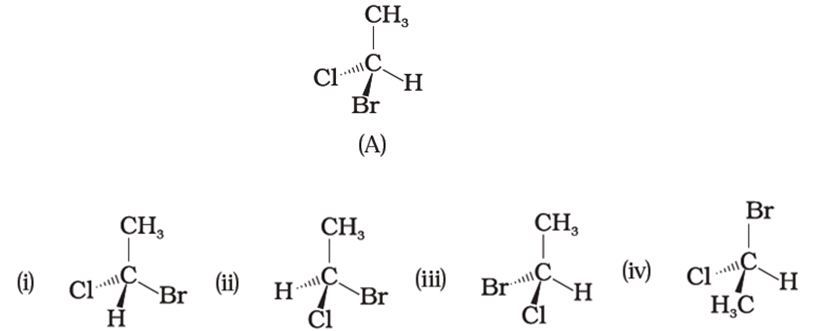

Answer : Option A
Explaination / Solution:
No Explaination.
Q9. Order of the photochemical reaction occurring between hydrogen and chlorine is
Answer : Option B
Explaination / Solution:
Photochemical reaction between and is 0 zero wrt to reactants.
Q10. Whipped cream is an example of
Answer : Option D
Explaination / Solution:
Whipped cream is example of foam.
