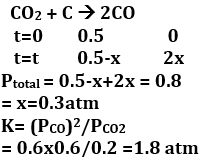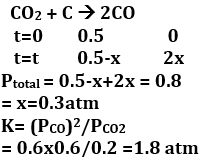Chemistry - Online Test
Q1. Liquid – liquid sols are known as
Answer : Option B
Explaination / Solution:
Liquid in liquid sols are emulsions
Q2. The interaction energy of London force is inversely proportional to sixth power of the distance between two interacting particles but their magnitude depends upon
Answer : Option A
Explaination / Solution:
Its magnitude depends on the polarisability of interacting particles.
Q3. Conjugated protein is.
Answer : Option B
Explaination / Solution:
Conjugated protein is phosphoprotein.
Q4. n – propyl bromide on treating with alcoholic KOH produces
Answer : Option A
Explaination / Solution:
Alkenes can be prepared from alkyl halides by treatment with alcoholic solution of caustic potash (KOH) at about 353-363 K. This reaction is known as dehydrohalogenation of alkyl halides.
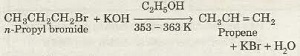
Q5. 35 mL of 0.1 M solution is used to neutralize 25 mL of barium hydroxide. The molarity of Barium hydroxide solution is
Answer : Option A
Explaination / Solution:
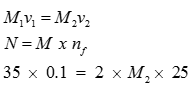
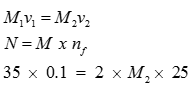
Q6. Esterification is the reaction of one of the below compounds with alcohol
Answer : Option A
Explaination / Solution:
Carboxylic acid and alcohol reacts to form an ester.
Q7. There are ____ L in 0.05 ml?
Answer : Option D
Explaination / Solution:
Since , 1L
= 1000mL
or , 1mL
=.
0.05mL
=
= 0.00005 L .
Q8.
The compound formed as a result of oxidation of ethyl benzene by is
Answer : Option C
Explaination / Solution:
Alkyl benzene on oxidation with give benzoic acid.
Q9. Leaching of gold is done by
Answer : Option A
Explaination / Solution:
Cyanide forms soluble complex with the gold.
Q10.
A vessel at 1000 K contains with a pressure of 0.5 atm. Some of the is converted into CO on the addition of graphite. If the total pressure at equilibrium is 0.8 atm, the value of K is
Answer : Option A
Explaination / Solution: