Chemistry - Online Test
Q1. Tetraaminecopper(II) ion is a square planar complex with one unpaired electron. According to valence bond theory the hybrid state of copper should be
Answer : Option C
Explaination / Solution:
Tetraaminecopper(II) ion is square planar so hybridization is dsp2
Q2. Artificial sweetening agents are given to diabetics because
Answer : Option A
Explaination / Solution:
Artificial sweetening agents are metabolized and hence don’t add calories.
Q3. The product formed in Aldol condensation is
Answer : Option C
Explaination / Solution:
In aldol we get beta hydroxyl aldehyde/Ketone.
Q4. Saturated solution of is used to make 'salt bridge' because
Answer : Option C
Explaination / Solution:
Saturated solution of is used in salt bridge because of same mobility o f and
Q5. The volume of gas is reduced to half from its original volume. The specific heat will be
Answer : Option C
Explaination / Solution:
Specific heat will remain constant.
Q6. In ion the formal charge on the oxygen atom of P–O bond is
Answer : Option D
Explaination / Solution:
Formal charge on the oxygen atom of P–O bond is – 1
Q7. Decomposition of benzene diozonium chloride by using Cu2Cl2/HCl to form chlorobenzene is
Answer : Option C
Explaination / Solution:
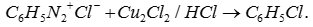
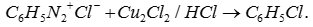
Q8. The reducing power of a metal depends on various factors. Suggest the factor which makes Li, the strongest reducing agent in aqueous solution.
Answer : Option A
Explaination / Solution:
High hydration energy of Li makes it’s a very good reducing agent. Its hydration energy compensates the high ionization energy.
Q9. Which of the following compounds contain all the carbon atoms in the same hybridisation state?
Answer : Option A
Explaination / Solution:
In A all Carbon atoms are sp hybridized.
Q10. For an endothermic reaction, the minimum value for the energy of activation in terms of will be
Answer : Option C
Explaination / Solution:
>
