Chemistry - Online Test
Q1. The above can be aptly described as
Answer : Option D
Explaination / Solution:
Isoelectronic species are elements or ions that have the same, or equal number of electrons. Although isoelectronic species have the same number of electrons, they are different in their physical and chemical properties.
Q2. Energy of an electron in stationary state is given by the formula
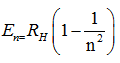
where n=1,2,3....... . In this equation, is called
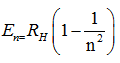
where n=1,2,3....... . In this equation, is called
Answer : Option D
Explaination / Solution:
The Rydberg constant, symbol R∞ for heavy atoms or RH for hydrogen, named after the Swedish physicist Johannes Rydberg, is a physical constant relating to atomic spectra, in the science of spectroscopy.
Q3. Which of these was an attempt by world governments to specifically address the problem of global warming?
Answer : Option B
Explaination / Solution:
The Kyoto Agreement was an attempt by world governments to specifically address the problem of global warming.
Q4. Vitamin B12 contains
Answer : Option C
Explaination / Solution:
Vitamin B12 containsCo3+
Q5.
The peroxide that gives H2O2 on treatment with dilute is
Answer : Option D
Explaination / Solution:
Barium peroxide react with sulfuric acid to produce hydrogen peroxide and barium sulfate. 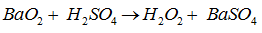
Q6. In Hell – Volhard Zelinsky reaction, halogen reacts with
Answer : Option B
Explaination / Solution:
Alpha Hydrogen containing carboxylic acids undergo HVZ reaction.
Q7. Which among the following will not show geometrical isomerism?
Answer : Option A
Explaination / Solution:
[Co(en)3]3+ all ligands are same its symmetrical so doesnot exists as geometrical isomers.
Q8. Aspirin is not administered for
Answer : Option A
Explaination / Solution:
For blood clotting aspirin is not taken.
Q9. For the reaction, , what are the signs of and ?
Answer : Option B
Explaination / Solution:
As in this cas e bond formation between two Cl atom is taking place so energy will be released so ΔH<0 and since 2 atoms are combining to form 1 molecule randomness decreases so ΔS<0.
Q10. Three electrolytic cells A,B,C containing solutions of ZnSO4, AgNO3 and CuSO4, respectively are connected in series. A steady current of 1.5 amperes was passed through them until 1.45 g of silver deposited at the cathode of cell. How long did the current flow? What mass of copper and zinc were deposited?
Answer : Option C
Explaination / Solution:
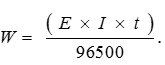
And mass of Zn and Cu deposited will be in ratio of their equivalent mass.
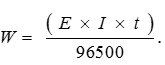
And mass of Zn and Cu deposited will be in ratio of their equivalent mass.
