Chemistry - Online Test
Q1. Which has highest freezing point at 1 atm?
Answer : Option A
Explaination / Solution:
Sugar solution has i=1 so ΔTf minimum so Tf will be maximum.
Q2. Using the standard electrode potential, find out the pair between which redox reactions is not feasible.
Answer : Option D
Explaination / Solution:
As for this couple is negative so this couple is not possible.
Q3. In Pyridine, preferred site of nucleophilic substitution is one of the following positions
Answer : Option B
Explaination / Solution:
Due to resonance, this position is electron rich and steric hindrance will be least.
Q4. What is the mass of 1 mole of O2 ?
Answer : Option B
Explaination / Solution:
Calculations:
Mass of 1mole of O2
= the mass of 6.022 x 1023 molecules of O2 ' ( as per Avogadro's law )
= molar mass of O2 in grams ( ie. gram molar mss )
= 32 .0 g
Q5. At a particular temperature and atmospheric pressure, the solid and liquid phases of a pure substance can exist in equilibrium. Which of the following term defines this temperature?
Answer : Option B
Explaination / Solution:
Normal melting both the phases liquid and solid exist in equilibrium.
Q6. The non-aromatic compound among the following is
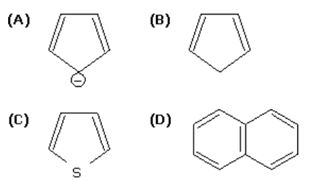

Answer : Option B
Explaination / Solution:
The compound B does not satisfy the (4n+2)pi electron rule and is therefore not an aromatic compound.
Q7. In the extraction of nickel by Mond’s process, the metal is obtained by
Answer : Option A
Explaination / Solution:
Ni is first reacted with CO to form Nickel tetracarbonyl which is volatile. From this complex Nickel is extracted by thermal decomposition.
Q8. The structure of ClF3 is
Answer : Option B
Explaination / Solution:
CN=0.5(V+M-C+A)
For ClF3 . CN= 5 so hyb is sp3d . Structure is trigonal bipyramidal.
Q9. At what temperature range do most of the metals become superconductors?
Answer : Option C
Explaination / Solution:
2K – 5K temperature metal behaves as superconductors.
Q10. In vulcanisarion of rubber,
Answer : Option D
Explaination / Solution:
In vulcanization of rubber sulphur crosslinking is introduced.
