Chemistry - Online Test
What is the correct order of reactivity of alcohols in the following reaction?
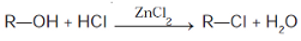
The mixture of HCl and ZnCl2 is called the Lucas Reagent. Secondary and tertiary alcohols react via the SN1 mechanism with the Lucas reagent. The ZnCl2 coordinates to the hydroxyl oxygen, and this generates a far superior leaving group.
When alcohols react with a hydrogen halide, a substitution occurs, producing an alkyl halide and water:
Scope of Reaction: The order of reactivity of alcohols is 3° > 2° > 1°
Using the standard electrode potential of redox couples given below find out which of the following is the strongest oxidising agent.
E0 values: Fe3+/Fe2+ = +0.77; I2/I-(s) = +0.54; Cu2+/Cu = +0.34; Ag+/Ag = +0.80
Choose the most appropriate option to complete the statement -
" Whenever there is a rearrangement of atoms which makes or breaks chemical bonds ------------------------ "
2- Methyl -2 - pentene when treated with ozone forms an unstable ozonide intermediate . The ozonide when treated with yields an aldehyde (propionaldehyde ) and a ketone ( acetone ) as the final products The conversion reactions are represented as below.
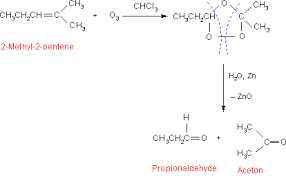
Thus , the compound A is (2-Methyl -2 -pentene )
