Chemistry - Online Test
Q1. Alkyl benzenesulphonate, an anionic detergent is widely used in
Answer : Option D
Explaination / Solution:
It is used in house hold detergents.
Q2. Given = –-92.4 kJ What is the standard enthalpy of formation of gas?
Answer : Option B
Explaination / Solution:
Standard Enthalpy of formation of NH3 is the energy change that take place when 1 mole of NH3 is formed from elements in standard state. For 2 moles of ammonia formation energy= –-92.4 kJ . For 1 mole energy =0.5 x–-92.4 kJ = -46.2 kJ
Q3. How much electricity is required in coulomb for the oxidation of 1 mol of to ?
Answer : Option C
Explaination / Solution:
For 1 mol to 2 mol of electrons are required.
Q4. Which one of the following is paramagnetic?
Answer : Option C
Explaination / Solution:
The number of electrons in Nitrogen are 7 and in oxygen are 8. Hence the number of electrons that would be present in the molecular orbitals in NO are 7 + 8 = 15. As the number of electrons are odd, all the electrons in NO molecule cannot be paired. Hence, a single electron would be present in a π*2p orbital. Therefore NO is an odd electron species and the gas is hence paramagnetic due to the presence of unpaired electron.
Q5. Freons diffuses unchanged into the stratosphere. It can
Answer : Option D
Explaination / Solution:
Freons disturbs the natural ozone balance and initial free radical rxn.
Q6. Choose the correct order of increase in ionic size of alkaline earth metals in the options given below
Answer : Option D
Explaination / Solution:
As we move down the group ionic radii increases.
Q7. Aniline is separated from aniline-water mixture using following technique:
Answer : Option B
Explaination / Solution:
Steam distillation is a special type of distillation(separation process) for temperature sensitive materials like natural aromatic compounds.It was a popular laboratory method for purification of organic compounds..
Q8.
In the catalyzed decomposition of benzene diazonium chloride,

Half life period is found to be independent of the initial concentration of the reactant. After 10 min, the volume of gas collected is 10 L and after the reaction is complete, it is 50 L. Hence, the rate constant of the reaction(in min ) is

Half life period is found to be independent of the initial concentration of the reactant. After 10 min, the volume of gas collected is 10 L and after the reaction is complete, it is 50 L. Hence, the rate constant of the reaction(in min ) is
Answer : Option C
Explaination / Solution:
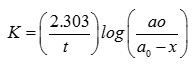
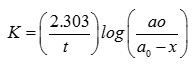
Q9. Which type of a property is the Brownian movement of colloidal solution?
Answer : Option C
Explaination / Solution:
Movement is always a mechanical property.
Q10. The van der Waals Equation adjusts the measured volume
Answer : Option A
Explaination / Solution:
nb is subtracted.
