Chemistry - Online Test
Q1. Calculate the total number of electrons present in 1.4 g of dinitrogen gas.
Answer : Option C
Explaination / Solution:
molesofN2=1.428=0.05molAnd1moleofN2=6.022×1023moleculesofN2And1moleculeofN2has14electronstotalnumberofelectronsin1.4gofN2=0.5×6.022×1023×14=4.214×1023
Q2. RNA guides the biosynthesis of
Answer : Option D
Explaination / Solution:
RNA guides the biosynthesis of proteins.
Q3. Ketones are reduced to the corresponding alcohols by catalytic hydrogenation to form
Answer : Option D
Explaination / Solution:
Carboxylic acids, esters, and acid halides can be reduced to either aldehydes or a step further to primary alcohols, depending on the strength of the reducing agent; aldehydes and ketones can be reduced respectively to primary and secondary alcohols.
Q4.
On increasing the pressure, in which direction will the gas phase reaction proceed to re-establish equilibrium is predicted by applying the Le Chatelier’s principle. Consider the reaction. (g) Which of the following is correct, if the total pressure at which the equilibrium is established, is increased without changing the temperature?
Answer : Option D
Explaination / Solution:
is independent of P change ,only the position at which equilibrium will be established will differ.
Q5. Ethylene glycol is added to water as antifreeze. It will
Answer : Option B
Explaination / Solution:
Fact.
Q6. The oxidation number of an element in a compound is evaluated on the basis of certain rules. Which of the following rules is not correct in this respect?
Answer : Option C
Explaination / Solution:
as oxygen is more electronegative than Cl,Br and I. So they have positive oxidation state.
Q7. When pyridine is treated with a mixture of nitric and sulfuric acids, the major product is:
Answer : Option B
Explaination / Solution:
In pyridine EAS take splace at 3 position.
Q8. The molarity of NaOH in a solution prepared by dissolving its 4.0 g in enough water to form 250 mL of the solution is ,
Answer : Option C
Explaination / Solution:
Since , Molarity
=
substituting the given values , we get -
Molarity ( M )
=
=0.4M
* moles of solute ie. NaOH = mole
.= 0.1mol
Q9. Identify B and D in the following sequence of reactions.
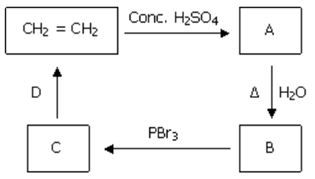

Answer : Option C
Explaination / Solution:
Ethanolis formed on addition of water, alcoholic KOH results in formation of alkene.
Q10. At 1123 K temperature in the blast furnace , CO reduces
Answer : Option B
Explaination / Solution:
At this Temp range free energy for the process is negative.
