Chemistry - Online Test
Q1. The compound [Cr(H2O)6]Cl3 and [Cr(H2O)4Cl2]Cl.2H2O represent
Answer : Option B
Explaination / Solution:
There is exchange of 4H_2O$ molecules inside and outside the coordination sphere.
Q2. Enthalpies of formation of CO(g), , and are -110, –-393, 81 and 9.7 kJ respectively. Find the value of for the reaction:
Answer : Option C
Explaination / Solution:
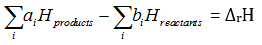
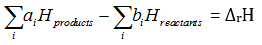
Q3. Scum formation can be prevented by
Answer : Option D
Explaination / Solution:
Scum formation happens in hard water. It ca be avoided by adding water softners or by using detergents.
Q4. How much electricity in terms of Faraday is required to produce 40.0 g of Al from molten ?
Answer : Option C
Explaination / Solution:
Moles of Al= 40/27. For 1 mol of Al deposition 3F charge is required.
Q5. Among the following the maximum covalent character is shown by the compound
Answer : Option A
Explaination / Solution:
Few ionic bonds have partial covalent characteristics which were first discussed by a scientist, Kazimierz Fajans in 1923. He gave the rule to predict whether a chemical bond will be covalent or ionic which is now commonly known as Fajan’s rule. The rule can be stated as:
- Size of the ion: Smaller the size of cation, larger the size of anion, greater is the covalent character of the ionic bond.
- Charge of Cation: Greater the charge of cation, greater is the covalent character of the ionic bond.
- Electronic configuration- For cations with same charge and size, the one, with (n-1)dnns0 which is found in transition elements have greater covalent character than the cation with ns2np6 electronic configuration, which is commonly found in alkali or alkaline earth metals.
Keeping these rules in mind, we can say that AlCl3 has high covalent character.
Q6. Dichloromethane (Methylene chloride) is
Answer : Option B
Explaination / Solution:
CHCl2 is organic solvent
Q7. Choose the correct order of increase in electopositivity of alkaline earth metals in the options given below
Answer : Option D
Explaination / Solution:
As we move down the group size increases so IE value decreases thus electropositivity also decreases.( shielding effect also decreases as we go down the Group)
Q8. The technique that is extensively used to separate mixtures into their components, purify compounds and also to test the purity of compounds is
Answer : Option A
Explaination / Solution:
Chromatography is used to separate mixtures of substances into their components. All forms of chromatography work on the same principle. They all have a stationary phase (a solid, or a liquid supported on a solid) and a mobile phase (a liquid or a gas).
Q9. The ionic reactions are generally very fast because
Answer : Option B
Explaination / Solution:
Ionic reactions does not involve bond breaking therefore they are fast.
Q10. Which of the following processes does not involve a catalyst?
Answer : Option A
Explaination / Solution:
Thermite process doesn’t require a catalyst.
