Chemistry - Online Test
Q1. The impurities associated with ore is known as
Answer : Option C
Explaination / Solution:
Gangue are the unwanted particles present with ores.
Q2. in the Thomson Model of Atom
Answer : Option D
Explaination / Solution:
- J. J. Thomson, who discovered the electron in 1897, proposed the plum pudding model of the atom in 1904 before the discovery of the atomic nucleus in order to include the electron in the atomic model.
- In Thomson's model, the atom is composed of electrons surrounded by a soup of positive charge to balance the electrons' negative charges, like negatively charged "plums" surrounded by positively charged "pudding".
- The 1904 Thomson model was disproved by Hans Geiger's and Ernest Marsden's 1909 gold foil experiment.
Q3. Tropospheric and stratospheric pollution are actually the components of :
Answer : Option C
Explaination / Solution:
Tropospheric and stratospheric pollution is the pollution associated with tropospheric and stratospheric layers of atmosphere so they are components of atmospheric pollution.
Q4. Which among the following elements does not belong to first transition series?
Answer : Option B
Explaination / Solution:
Ag belongs to second transition series.
Q5. The following compound is called
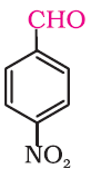

Answer : Option A
Explaination / Solution:
This is 4 – nitrobenzenecarbaldehyde and 4 – nitrobenzaldehyde
Q6. The coordination number of Cr in [Cr(NH3)3(H2O)3]Cl3 is
Answer : Option D
Explaination / Solution:
Coordination number of the central ion is equal to no. of coordination bonds formed with surrounding ions.
Q7. The three isotopes of hydrogen in terms of chemical properties show one of the following properties
Answer : Option D
Explaination / Solution:
Isotopes difference in their rate of reaction.
Q8. The narrow spectrum antibiotic is
Answer : Option C
Explaination / Solution:
Penicillin G is narrow spectrum antibiotic.
Q9. Standard electrode potential are measured with the concentrations of all the species involved in a half-cell is
Answer : Option A
Explaination / Solution:
Under standard condition concentration of every species is taken as 1.
Q10. The number of dots around the Lewis symbols for the elements represent
Answer : Option A
Explaination / Solution:
Gilbert N. Lewis is widely known for his use of simple symbolic representations of elements that show valence electrons as dots.
The Lewis electron-dot symbols focus on the electrons in the highest principal energy level in the atom, the valence electrons.
After all, these are the electrons that participate in chemical reactions. Lewis electron-dot symbols work well for the representative elements.
