Chemistry - Online Test
Q1. Which of the following statements about Hydrogen bond incorrect?
Answer : Option C
Explaination / Solution:
Hydrogen atom covalently bonded to highly electronegative atom such as N,O experience electrostatic field of another highly electronegative atom due to which a partial positive charge is developed on H atom.
Q2. The following amine can be classified as (C2H5)2NH
Answer : Option B
Explaination / Solution:
Nitrogen is attached is two carbons, so this is asecondary amine.
Q3.
Give IUPAC names of the following compound:
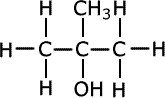
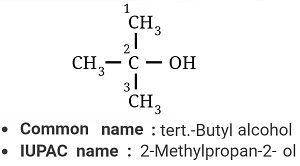
Answer : Option C
Explaination / Solution:
Longest chain has three carbons and at 2nd position both –OH and –CH3 group are attachted.
Q4. Which of these is not a characteristic of aromatic Hydrocarbons?
Answer : Option B
Explaination / Solution:
aromatic hydrocarbon are not open chained. They are ring structures.they contain one or more benzene rings either fused or isolated in their molecules.
Q5. For ideal solution the enthalpy of mixing of the pure components to form the solution is
Answer : Option D
Explaination / Solution:
For ideal solution there is no evolution or absorption of heat on mixing.
Q6. Which among the following is an example of network solid?
Answer : Option D
Explaination / Solution:
Carbon is network solid due to its tendency to form Catination.
Q7. Mendeleev proposed that some of the elements were still undiscovered and, therefore, left several gaps in the table. He left the gap under aluminium and a gap under silicon, and called these elements Eka-Aluminium and Eka-Silicon. These were later discovered and named
Answer : Option C
Explaination / Solution:
Mendeleev arranged the elements according to their atomic weights. He corrected the atomic weights of few exisiting elements to explain their chemical and physical behaviour. Moreover, he felt that few elements are still to be discovered, so he left empty spaces for them in his periodic table. Two such elements whose properties he predicted; and were later discovered are Gallium and Germanium respectively.
Q8. Polythene is not one of the following
Answer : Option B
Explaination / Solution:
Polymer is not semi synthetic. It is synthetic.
Q9. The maximum covalency of nitrogen is
Answer : Option C
Explaination / Solution:
N has maximum covalency of 4 since only four (one s and three p) orbitals are available for bonding. d orbitals are not available for bonding.
Q10. According to first law of thermodynamics ΔU = q + w, where
Answer : Option B
Explaination / Solution:
In thermodynamics q=+ve when heat is supplied to the system. And w=+ve when work is done on the system while when work is done by the system w is taken as –ve.
