Chemistry - Online Test
Q1. What is the correct order of decreasing stability of the following cations.
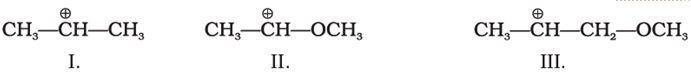
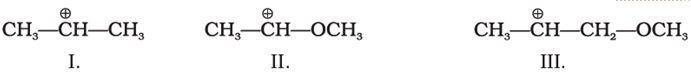
Answer : Option B
Explaination / Solution:
II is very stable because +ve charge is dispersed by +R effect of
Q2. What is the oxidation state of K in
Answer : Option C
Explaination / Solution:
K has +1 oxidation state. "The potassium atom losses one electron when forming compound as it has one electron on it's valence shell".
Q3. The radioactive isotope used in determining the age of organic substances is
Answer : Option A
Explaination / Solution:
The ratio of Carbon -12 to Carbon-14 at the moment of death is the same as every other living thing., but Carbon-14 decays and is not replaced. The carbon-14 decays with its half-life of 5770 years., while the amount of Carbon-12 remains constant in the sample. By looking at the ratio of Carbon-12 to Carbon-14 in the sample and comparing it to the ratio in a living organism, it is possible to determine the age of a formerly living thing fairly precisely.
Q4. In which of the following systems, the dispersed phase and dispersion medium are both solid?
Answer : Option A
Explaination / Solution:
It is a Sol type of colloid.
Q5. The deviation from ideal behavior can be measured in terms of compressibility factor Z, which is
Answer : Option A
Explaination / Solution:
Z=PV/nRT, is a correction factor which describe the deviation of real gas from ideal gas behaviour. it is defined as the ratio of the molar volume of a gas to the molar volume of an ideal gas at the same temperature and pressure.
Q6. A particular ratio of product to reactant helps in predicting the direction in which a given reaction will proceed at any stage. is called
Answer : Option C
Explaination / Solution:
Reaction Quoitennt (Q) =K ( equilibrium) Q>K (backward Reaction) Q
Q7. One of the following hassweet taste
Answer : Option A
Explaination / Solution:
Glucose, glycine have sweet taste.
Q8. Lucas reagent is
Answer : Option A
Explaination / Solution:
Lucas reagent is a solution of anhydrous zinc chloride (Lewis acid) in concentrated hydrochloric acid. It is used as a reagent to test alcohols and classify them in accordance to their reactivity. The reaction is a substitution reaction where the chloride of the zinc chloride gets replaced by the hydroxyl group of the alcohol.
Q9. The preparation of alkynes from vicinal dihalides is an example of---------- ?
Answer : Option C
Explaination / Solution:
Elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either one or two steps .When the starting dihalide is a vicinal dihalide , alkyne is obtained by two successive elimination reactions.
Example :
Vicinal dihalide, on treatment with forms a monosubstituted alkene Vinyl bromidewhich when reacted with sodium amide , forms ethyne (ie. an alkyne ). The steps of conversion are ,
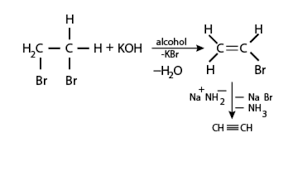
Q10. The molal depression constant depends upon
Answer : Option B
Explaination / Solution:
Kf is characteristic of solvent.
