Chemistry - Online Test
Q1. Which of the following pollutant gases naturally occurs in the environment :
Answer : Option D
Explaination / Solution:
SO2 is a naturally occurring pollutant gas with an approximate atmospheric concentration of 1 ppm. It is produced naturally by volcanic activities and burning of fossil fuels contaning sulphur.
Q2. Which of the following metals have only one oxidation state?
Answer : Option B
Explaination / Solution:
Al is not a d block element hence it shows only one oxidation state. While rest all others are d block elements which shows variable oxidation state.
Q3. One of the following is used for the separation and purification of aldehydes
Answer : Option A
Explaination / Solution:
NaHSO3 is used for purification of aldehydes.
Q4. According to the law of conservation of mass, a balanced chemical equation
Answer : Option C
Explaination / Solution:
According to law of conservation of mass
total mass of reactants
= total mass of product
∴ the number . of atoms of each element in reactants
= number . of atoms of that element in products
Q5. Metal hydrides are used for hydrogen storage because
Answer : Option B
Explaination / Solution:
Metal hydrides (MHx) are the most technologically relevant class of hydrogen storage materials because they can be used in a range of applications including neutron moderation,electrochemical cycling, thermal storage, heat pumps, and purification/separation.
The metal hydrides such as intermetallic alloys and solid solutions have interstitial vacancies where atomic hydrogen is absorbed via an exothermic reaction; however, by endothermic path, the metal hydride desorbs the hydrogen reversibly at ambient to moderate temperatures.
Q6. Hormones relating to birth control are
Answer : Option C
Explaination / Solution:
Hormones related to birth control are Estrogen and progesterone.
Q7. Which is not true about a ligand?
Answer : Option B
Explaination / Solution:
Multidentate ligand always act as chelating Ligand
Q8. A secondary cell after use ________ recharged by passing current through it in the ________ so that it can/can’t be used again.
Answer : Option B
Explaination / Solution:
Secondary cell can be recharged and reused.
Q9. In the formation of hydrogen molecule, overlapping of atomic orbitals which results in the pairing of electrons. These are:
Answer : Option A
Explaination / Solution:
overlapping of atomic orbitals having electrons of opposite spin take palce in the formation of molecule.
Q10. 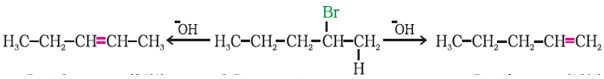
In the reaction Pent – 2 – ene. 2 – Bromopentane. Pent – 1 – ene 2 – Bromopentane on heating with alcoholic KOH, forms two compounds: Pent – 1 – ene and Pent – 2 – ene. Which one of the following statements is true
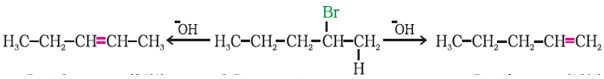
In the reaction Pent – 2 – ene. 2 – Bromopentane. Pent – 1 – ene 2 – Bromopentane on heating with alcoholic KOH, forms two compounds: Pent – 1 – ene and Pent – 2 – ene. Which one of the following statements is true
Answer : Option D
Explaination / Solution:
Pent-2-ene is major product as ore substituted alkene is formed in major quantity according to zaitsev rule.
