Chemistry - Online Test
Q1. the rates of transfer of molecules from ice into water and of reverse transfer from water into ice are equal at atmospheric pressure and 273 K. Both the processes occur simultaneously and at the same rate so that the amount of ice and water remains constant. This process is called
Answer : Option C
Explaination / Solution:
Ice and water kept in a perfectly insulated thermos flask at 273K and the atmospheric pressure are in equilibrium state and the system shows interesting characteristic features. The mass of ice and water do not change with time and the temperature remains constant. However, the equilibrium is not static. The intense activity can be noticed at the boundary between ice and water. Molecules from the liquid water collide against ice and adhere to it and some molecules of ice escape into liquid phase. There is no change of mass of ice and water, as the rates of transfer of molecules from ice into water and of reverse transfer from water into ice are equal at atmosphere pressure and 273K.
Q2. dipole-dipole forces act between molecules that have
Answer : Option B
Explaination / Solution:
dipole dipole forces act between molecules that have a permanent electrical dipole moment,the partial charge of one molecule get attracted to the opposite charge of other molecule
Q3. The following amine can be classified as
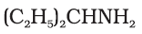
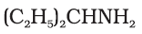
Answer : Option C
Explaination / Solution:
This is primary amine, because the Nitrogen of amine is only connected to one carbon.
Q4. Give IUPAC names of the following compound:
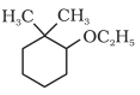

Answer : Option A
Explaination / Solution:
Lowest set of locants: The lowest set of locants is defined as the set that, when compared term by term with other locant sets, each cited in order of increasing value, has the lowest term at the first point of difference.
With regard to numbering of locants, simple prefixes (simple substituent groups consisting of just one part that describes an atom, or group of atoms as a unit, for example methyl and ethoxy) are considered together with equal seniority:
Therefore, the given compound is named as 2-ethoxy-1,1-dimethylcyclohexane rather than 1-ethoxy-2,2-dimethylcyclohexane since the locant set ‘1,1,2’ is lower than ‘1,2,2’.
Q5. in cycloalkanes carbon atoms form
Answer : Option D
Explaination / Solution:
Cycloalkanes contain single bond between the carbon atoms,they form ring structures and have the general formula CnH2n
Q6. Which law shows relation between pressure and solubility of a gas in solvent
Answer : Option B
Explaination / Solution:
Henry’s Law states that as the partial pressure of gas increases over liquid solubility of gas also increases.
Q7. Which among thThey do not show long range ordere following is not true for crystalline solid?
Answer : Option A
Explaination / Solution:
In Crystalline solids , the atomic pattern repeats itself after long distance. Therefore they have long range orders.
Q8. Mendeleev relied on the similarities in the empirical formulas and properties of the compounds formed by the elements. He realized that some of the elements did not fit in with his scheme of classification if the order of atomic weight was strictly followed. He therefore
Answer : Option D
Explaination / Solution:
Mendeleev placed the elements with similar properties together
Q9. cellulose acetate (rayon) and cellulose nitrate are __________ polymers
Answer : Option A
Explaination / Solution:
It is semi synthetic polymer.
Q10. In which of the following compound, nitrogen shows the oxidation state of +5?
Answer : Option A
Explaination / Solution:
Oxidation state of O is -2 and H is +1. Let the oxidation state of N is x.
(+1) +x + 3(-2) = 0
x = +5
