Chemistry - Online Test
Q1. The adsorbent used to adsorb the dye particles in the dying industry is
Answer : Option B
Explaination / Solution:
Alum is used in dying industries.
Q2. Viscous force is
Answer : Option A
Explaination / Solution:
Viscous force is directly proportional to area and velocity gradient.
Q3.
We know that the relationship between and is What would be the value of Δngas for the reaction
Answer : Option B
Explaination / Solution:
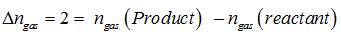
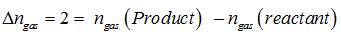
Q4. Which of the following is most soluble in water?
Answer : Option B
Explaination / Solution:
As they have polar interactions in water hence are soluble.
Q5. The reaction

is known as

is known as
Answer : Option A
Explaination / Solution:
Williamson’s synthesis: When an alkyl halide reacts with sodium alkoxide, ether is formed. This reaction is known as Williamson’s synthesis. The reaction generally follows SN2 mechanism for primary alcohols.
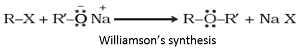
Q6. Which among the following is an example of liquid in solid?
Answer : Option A
Explaination / Solution:
Hydrated salts have water molecule of hydration as solute.
Q7. Heating a(n) __________ results in a Cope elimination.
Answer : Option D
Explaination / Solution:
Heating of Amine oxide result in cope elimination cis elimination.
Q8. Assign oxidation number to S in
Answer : Option D
Explaination / Solution:
Oxidation state of S in KAl(SO4)2 is +6
Q9.
Consider the chemical reaction gven as ,
-----CO (g) + ----- (g) → ----- (l) + ----- .
This equation can be balanced by inserting the following in blank spaces
Answer : Option B
Explaination / Solution:
The balanced equation for the given equation is ,
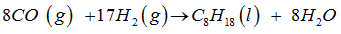
hence , the coefficients ( 8 ,17 , 1, 8 ) are inserted on blank spaces ( from LHS to RHS )
Q10. Which of the following turns lead acetate paper black?
Answer : Option D
Explaination / Solution:
Pb2++S2−→PbS(black)
