Chemistry - Online Test
Q1. Water behaves as an amphoteric substance because
Answer : Option A
Explaination / Solution:
An amphoteric compound is a compound that can act either as an acid, or as a base in a chemical reaction.
A first example of water's amphoteric character is the fact that it can self-ionize to a small extent. A water molecule is capable of donating one of its protons (H+) to a neighbouring water molecule, forming the hydroxyde and hydronium ions.
Q2. The artificial sweetener stable at high temperature is
Answer : Option C
Explaination / Solution:
Sucralose is non nutritive artificial sweetner.
Q3. In the formation of complex entity, the central atom/ion acts as
Answer : Option A
Explaination / Solution:
Central Metal ion behave as a Lewis Acid while Ligand act as a lewis Base.
Q4. Correct arrangement of Al, Cu, Fe, Mg and Zn in the order in which they displace each other from the solution of their salts is
Answer : Option B
Explaination / Solution:
Reactivity series.
Q5.
In , molecule below the dipole moment is zero although the B- F bonds are oriented at an angle of to one another. Net dipole moment in molecule is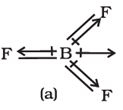
Answer : Option A
Explaination / Solution:
BF3 has symmetrical structure in which the three B-F bonds are oriented at an angle of 120° to one another. Also the three bonds lie in one plane and the dipole moments of these bonds cancel one another giving net dipole moment equal to zero.
Q6. Grignard reagent should be prepared under anhydrous conditions because
Answer : Option C
Explaination / Solution:
R—MgX is very reactive.
Q7. lithium halides are somewhat covalent because
Answer : Option D
Explaination / Solution:
Smaller is the size, greater is the polarization . so more is the covalent character.
Q8.
If an – functional group is present in an organic compound it is called amine. If a functional group is present in an organic compound then it is called
Answer : Option C
Explaination / Solution:
is amide functional group.
Q9. Collision theory is applicable to
Answer : Option C
Explaination / Solution:
Collision theory is only applicable to bimolecular reaction.
Q10. Adsorbent used for drying of air and controlling humidity in rooms is
Answer : Option A
Explaination / Solution:
Aluminia gel adsorbs humidity in air.
