Chemistry - Online Test
Q1. The following reaction is
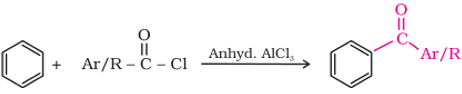

Answer : Option C
Explaination / Solution:
This is Friedal Craft reaction.
Q2. The calculation of masses or, (sometimes volumes also) of the reactants and the products involved in a chemical reaction is called
Answer : Option B
Explaination / Solution:
Stoichiometry is a method to express quantitative aspects of a chemical reaction .
Usually , the masses of reactants as well as those of products in a chemical reaction are calculated using corresponding balanced chemical equation .
It is convenient and hence desirable to calculate volumes of gaseous reactants and products .
Q3. presence of extensive hydrogen bonding between water molecules leads to
Answer : Option C
Explaination / Solution:
Water has high freezing point and boiling point because of strong intermolecular hydrogen bonding interactions.
Q4. Barbiturates are important class of
Answer : Option B
Explaination / Solution:
Barbiturates are important class of Tranquilisers.
Q5. KCl.MgCl2.6H2O is a
Answer : Option D
Explaination / Solution:
KCl.MgCl2.6H2O is double salt.
Q6. In corrosion a metal is ______ of electrons to oxygen and formation of oxides
Answer : Option D
Explaination / Solution:
Metals is oxidized by loss of electrons on corrosion.
Q7. in case of a heteronuclear molecule like HF, the shared electron pair between the two atoms gets displaced more towards fluorine since the electronegativity of fluorine is far greater than that of hydrogen. This results in the molecul
Answer : Option C
Explaination / Solution:
because of slight shifting of electron towards more electronegative element like F in HF brings a small amount of negative charge on F and positive charge on H which results in dipole moment.
Q8.
The dipole moment of chlorobenzene is lower than that of cyclohexyl chloride
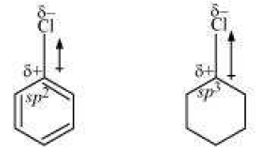 chlorobenzenecyclohexyl chloride
chlorobenzenecyclohexyl chloride
 chlorobenzenecyclohexyl chloride
chlorobenzenecyclohexyl chloride
Answer : Option D
Explaination / Solution:
Sp2 carbon is more electronegative than sp3 carbon.
Q9. The nitrogen containing organic compound, when heated with copper oxide in an atmosphere of carbon dioxide, yields:
Answer : Option C
Explaination / Solution:
The nitrogen containing organic compound, when heated with copper oxide in an atmosphere of carbon dioxide, yields free nitrogen
Q10. Lithium has most negative value,its reaction with water is less vigorous than that of sodium which has the least negative E0 value among the alkali metals. The reason is
Answer : Option D
Explaination / Solution:
Li due its small size have high Lattice energy value which compensates its high ionization energy so has most negative value
