Chemistry - Online Test
Q1.
What is M?
Answer : Option B
Explaination / Solution:
M is alkali metal. Alkali metals reacts with water vigorously to form hydroxide (OH-) and dihydrogen (H2)
Q2. A reaction whose order is different from the actual due to large excess concentration of one of the reactants is called
Answer : Option C
Explaination / Solution:
The concentration of the reactants used in excess remains unaltered during the course of the reaction.
Q3. Ammonia is adsorbed by
Answer : Option A
Explaination / Solution:
Charcoal act as an adsorbent.
Q4. behaviour of the gas becomes more ideal when
Answer : Option D
Explaination / Solution:
This question is based on understanding the concept of molecular interactions between molecule of real gases which is least at low P and High T condition. Thus under these conditions real gases behave as ideal.
Q5. The value of for a reaction is directly related to the thermodynamics of the reaction and in particular,
Answer : Option A
Explaination / Solution:
Δ G = -RTlnK
Q6. Protein is generally a polypeptide with _______ amino acid residues
Answer : Option A
Explaination / Solution:
In protein more than 100 amino acids combine to form proteins
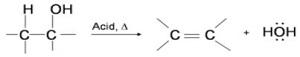
Q7. Alcohols undergo dehydration (removal of a molecule of water) to form alkenes on treating with
Answer : Option C
Explaination / Solution:
The dehydration reaction of alcohols to generate alkene proceeds by heating the alcohols in the presence of a strong acid, such as sulfuric or phosphoric acid, at high temperatures.
The required range of reaction temperature decreases with increasing substitution of the hydroxy-containing carbon:
- 1° alcohols: 170° - 180°C
- 2° alcohols: 100°– 140 °C
- 3° alcohols: 25°– 80°C
If the reaction is not sufficiently heated, the alcohols do not dehydrate to form alkenes, but react with one another to form ethers.
Q8. Majority of the reactions of alkynes are the examples of
Answer : Option D
Explaination / Solution:
Alkynes have two pi bonds,hence they are electron rich species.They attract electrophiles thereby undergoing electrophilic addition reactions.
Q9. Vapour pressure of a liquid is constant at constant temperature because
Answer : Option B
Explaination / Solution:
Liquid and vapours are in equilibrium.
Q10. Which of the following is a tertiary amine?
Answer : Option D
Explaination / Solution:
Benadryl have tertiary amine.