Chemistry - Online Test
Q1. According to quantum theoretical model of an atom each orbital is designated by three quantum numbers labelled as n, l and ml. These are referred to respectively as
Answer : Option B
Explaination / Solution:
Quantum numbers designate specific shells, subshells, orbitals, and spins of electrons. This means that they describe completely the characteristics of an electron in an atom, i.e., they describe each unique solution to the Schrödinger equation, or the wave function, of electrons in an atom.
There are a total of four quantum numbers: the principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic quantum number (ml), and the electron spin quantum number (ms). The principal quantum number, nn , describes the energy of an electron and the most probable distance of the electron from the nucleus. In other words, it refers to the size of the orbital and the energy level an electron is placed in. The number of subshells, or ll , describes the shape of the orbital. It can also be used to determine the number of angular nodes.
The magnetic quantum number, ml, describes the energy levels in a subshell, and ms refers to the spin on the electron, which can either be up or down.
Q2. Deforestation and burning of fossil fuels adds to the environmental pollution by :
Answer : Option C
Explaination / Solution:
Deforestation and burning of fossil fuels adds to the environmental pollution As it increases the amount of CO2 in the atmosphere which traps excessive heat and increases global temperature causing global warming.
Q3. Which of the following pairs of ions have same paramagnetic moment?
Answer : Option C
Explaination / Solution:
Cu2+, Ti3+ will have same unpaired electrons and hence same paramagnetic moment.
Q4. IUPAC name of the following compound is
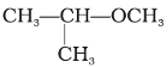

Answer : Option A
Explaination / Solution:
This is 1-methoxy-1-methylethane.
Q5. Molecular formula represents
Answer : Option D
Explaination / Solution:
Molecular formula is the exact no. of atoms present in a molecule of a compound.
Molecular formula of a compound is related with its empirical formula as ,
Mollecular formula = ( Empirical formula ) n
where n represents a positive integer .
Q6. Many d-block and f-block elements form
Answer : Option B
Explaination / Solution:
Hydrides are formed by many d-block and f-block elements when heated with hydrogen under pressure. The hydrides tend to be non-stoichiometric and they may be of variable composition.These hydrides have properties similar to those of parent metal and hence are known as metallic hydride.
Q7.
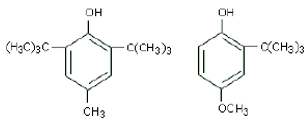
The compounds above are very good antioxidants. They are

The compounds above are very good antioxidants. They are
Answer : Option C
Explaination / Solution:
They are BHT and BHA.
Q8. The oxidation state of Ag in tollen’s reagent is
Answer : Option A
Explaination / Solution:
oxidation state of Ag in tollen’s reagent is +1.
Q9. In the primary batteries
Answer : Option C
Explaination / Solution:
Primary batteries cannot be recharged and reused.
Q10. the valence shell electron pair repulsion (vsepr) theory helps in the
Answer : Option A
Explaination / Solution:
Valence shell electron pair repulsion (VSEPR) theory is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. It is also named the Gillespie-Nyholm theory after its two main developers.
