Chemistry - Online Test
Q1. The product of the magnitude of the charge and the distance between the centres of positive and negative charge is called
Answer : Option D
Explaination / Solution:
A dipole moment is a measurement of the separation of two opposite electrical charges. Dipole moments are a vector quantity. The magnitude is equal to the charge multiplied by the distance between the charges and the direction is from negative charge to positive charge:
µ=q x r
Q2. Compounds containing carbon atoms joined together in the form of a ring as following are:
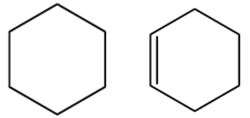

Answer : Option C
Explaination / Solution:
An alicyclic compound is an organic compound that is both aliphatic and cyclic. They contain one or more all-carbon rings which may be either saturated or unsaturated, but do not have aromatic character.
Q3. Which one of the following is not a chiral molecule?
Answer : Option C
Explaination / Solution:
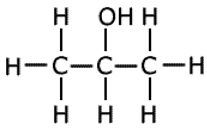 .this
is not chiral molecule because functional group carbon that is carbon
having alchohal group has 2 same groups on either side and there is
symmentry about this carbon that is why this is not chiral.
.this
is not chiral molecule because functional group carbon that is carbon
having alchohal group has 2 same groups on either side and there is
symmentry about this carbon that is why this is not chiral.
 .this
is not chiral molecule because functional group carbon that is carbon
having alchohal group has 2 same groups on either side and there is
symmentry about this carbon that is why this is not chiral.
.this
is not chiral molecule because functional group carbon that is carbon
having alchohal group has 2 same groups on either side and there is
symmentry about this carbon that is why this is not chiral.
Q4. The electronic configuration of Rn7s1 belongs to
Answer : Option D
Explaination / Solution:
Francium will fill orbitals all the way up to 7S (7th period) subshell whrer it will have half filled orbital : 7S1 .
Q5. The rate is independent of the concentration of the reactants in
Answer : Option A
Explaination / Solution:
−d[R]/dt = k[R]0
Q6. The colloidal solution of two immiscible liquids is called
Answer : Option A
Explaination / Solution:
Dispersed phase is liquid, dispersion medium is liquid.
Q7. Real gases show deviations from ideal gas law because
Answer : Option D
Explaination / Solution:
Real gases have intermolecular forces of interaction due to which there is a difference in the actual pressure values when compared to the ideal gases, the pressure is found to be less than the pressure of ideal gases under similar conditions, similar deviation occurs when the size of molecules is taken into account due to which volume correction is required.
Q8. For reactions involving gases, however, it is usually more convenient to express the equilibrium constant in terms of
Answer : Option C
Explaination / Solution:
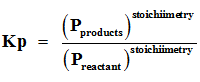
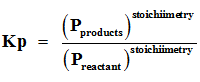
Q9. D – ribose and 2 – deoxy – D – ribose are
Answer : Option C
Explaination / Solution:
They are pentose sugar
Q10.
IUPAC name of the following compound is
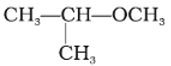
Answer : Option B
Explaination / Solution:
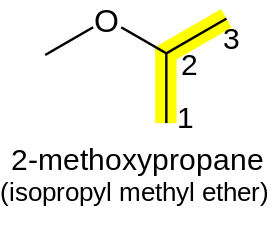
If the oxygen is not attached to the end of the main alkane chain, then the whole shorter alkyl-plus-ether group is treated as a side-chain and prefixed with its bonding position on the main chain. Thus CH3OCH(CH3)2 is 2-methoxypropane.
