Chemistry - Online Test
Q1. In a π (pi) bond formation, is necessary for one of the below criteria. Choose the appropriate one.
Answer : Option B
Explaination / Solution:
parallel orientataion of p orbitals are necessary for proper overlapping and formation of pie bond.
Q2. Fill up the blank with suitable word in the statement,
" Physical properties are those properties which are measured or observed--------------- changing the identity or the composition of the substance ."
Answer : Option D
Explaination / Solution:
A substance is identified by its composition , hence for measuring physical properties of a substance its identity must be retained , (or should not be changed ) Hence , the given statement ,
" Physical properties are those properties which are measured or observed without changing the identity or composition of the substance."
is correct.
Q3. For the same alkyl group, an alkyl bromide has a higher boiling point than alkyl fluoride because,
Answer : Option C
Explaination / Solution:
Vanderwall forces increases with increase in molecular mass and boiling point increases with vanderwall forces..
The main factor affecting the boiling point is the molecular weight, as can be pictured from the noble gases boiling point in the periodic table (the noble gases are those which have the weakest interactions due to their complete outer shell). But note that if you want to consider all the effects altogether, you must use Kelvins instead of Celsius. So going from 44°C to 88°C is not twice as much but a mere 14% increase.
The other factor affecting boiling point is the nature and strength of intermolecular interactions, usually in the order ionic > hydrogen bond > dipole-dipole > Van der Waals.
In the comparison of RF vs RBr boiling point, molecular weight is definitely in favor of higher boiling points for RBr, at least for small molecules. It is true that the C-F bond is much more ionized than C-Br so it will create stronger dipole-dipole interactions but not sufficiently to compensate for the effect of the mass increase.
Q4. The number of asymmetric carbon atom(s) in below figure is are
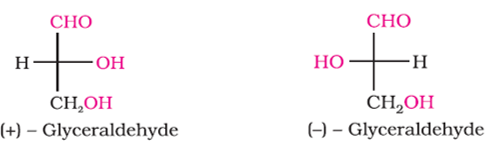
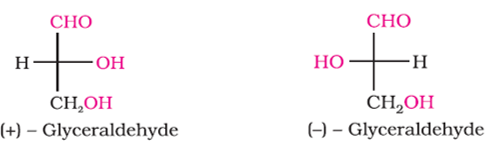
Answer : Option A
Explaination / Solution:
Only 1 asymmetric center.
Q5. Molecularity of a reaction
Answer : Option A
Explaination / Solution:
Molecularity means minimum no. of atoms molecules or ions which undergo collision for conversion of reactant to Product. So it is a whole no. Cannot be fractional or negative.
Q6. Metals like silver and copper can be obtained in the colloidal state by
Answer : Option D
Explaination / Solution:
Metal sols are prepared only by Bredig’s Arc method.
Q7. If we place solid iodine in a closed vessel, after sometime the vessel gets filled up with violet vapour. When equilibrium is attained, the intensity of colour will be
Answer : Option D
Explaination / Solution:
we place solid iodine in a closed vessel, after sometime the vessel gets filled up with violet vapour and the intensity of colour increases with time. After certain time the intensity of colour becomes constant and at this stage equilibrium is attained. Hence solid iodine sublimes to give iodine vapour and the iodine vapour condenses to give solid iodine. The equilibrium can be represented as, I2 (solid) ⇌ I2 (vapour)
Q8. The average Kinetic energy and Thermal energy are proportional to the
Answer : Option C
Explaination / Solution:
The average KE and Thermal energy are proportional to absolute Temperature.
The kinetic energy is the amount of motion in the atoms and molecules of a substance. The moleculess of the gas gain energy as the temperature increases as there is more motion of the molecules. As the temperature decreases the motion lessens until at absolute zero all motion (kinetic energy) stops.
Q9. The following reaction takes place in the presence of
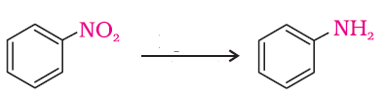

Answer : Option A
Explaination / Solution:
-NO2 group is reduced to –NH2 using H2/Pd.
Q10. C6H5OCH2CH3 is called
Answer : Option D
Explaination / Solution:
C6H5OCH2CH3 is ethyl phenyl ether or phenetole is an organic compound that is an ether. Ethyl phenyl ether has the same properties as some other ethers, such as volatility, explosive vapors, and the ability to form peroxides.
IUPAC name : Ethoxybenzene
Other names : Phenetole, Ethyl Phenyl Ether
