Chemistry - Online Test
Q1. Calcium and Magnesium belong to the category of
Answer : Option B
Explaination / Solution:
calcium and megnesium are 2nd group element of periodic table which are obtaine on earth in the form ore, their oxides and hydroxide are alkaline in nature so they are alkaline earth metals
Q2. The IUPAC name for
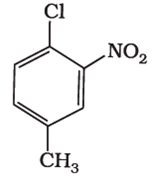

Answer : Option A
Explaination / Solution:
1-Chloro-4-methyl-2-nitrobenzene is the iupac name of the given compound.
Q3. Particles of compounds are
Answer : Option C
Explaination / Solution:
Compounds are made when,
atoms of two or more different elements combine together in fixed ratio to form molecules .(viz. water , ammonia , carbon- di- oxide glucose etc. ) .
Thus the particles of compound consists of molecules.
Q4. Yield of the following compound is high
Answer : Option D
Explaination / Solution:
This reaction is free radical reaction and 3o free radical is most stable.
Q5. Choose the appropriate answer(s) for the below representation from the options given
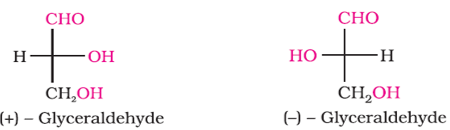
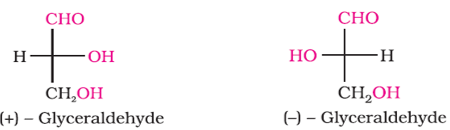
Answer : Option D
Explaination / Solution:
They are, enatiomers as 1 is R, other is S. Therefore are stereo isomers. And dextro and levo.
Q6.
What is the order of a reaction which has a rate expression ; Rate =
Answer : Option A
Explaination / Solution:
Total order of reaction = order wrt to A + order wrt to B.
Q7. Which one of the following is an example of micelle system?
Answer : Option B
Explaination / Solution:
Soap have hydrophobic tails and hydrophilic heads so they can act micelle.
Q8. During the process of transformation from liquid to vapour, the pressure exerted by the water molecules at a given temperature remains constant. This is called
Answer : Option D
Explaination / Solution:
The number of water molecules from the gaseous state into the liquid state also increases till the equilibrium is attained.
i.e.rate of evaporation = rate of condensation
H2O(l) ⇌ H2O (vap)
At equilibrium the pressure exerted by the water molecules at a given temperature remains constant and is called the equilibrium vapour pressure of water increases with temperature.
Q9. The intermolecular force primarily responsible for the condensed states of nonpolar substances is the
Answer : Option A
Explaination / Solution:
The intermolecular force primarily responsible for the condensed states of nonpolar substances is the dispersion force (or London force).
Q10. The following amine can be classified as
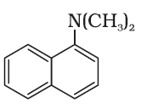

Answer : Option D
Explaination / Solution:
This is tertiary amine but since all the alkyl groups are attachted so it is mixed amine.
