Chemistry - Online Test
Q1. The rate law for the reaction is given by rate= k[RCl]. The rate for this reaction
Answer : Option C
Explaination / Solution:
Since order of reaction with respect to RCl is one so if conc of Rcl is halfed the rate of reaction will also become half.
Q2. The process of separation of colloids by passing through semi permeable membrane is called
Answer : Option C
Explaination / Solution:
Dialysis we use, salt solution of approximately same concentration outside the artificial blood capillary, so that urea comes out of the blood, but other useful substances like NaCl , blood cell etc does not. Blood is a colloid.
Q3. A plot of volume (V ) versus temperature (T ) for a gas at constant pressure is a straight line passing through the origin. The plots at different values of pressure are shown in Figure. Which of the following order of pressure is correct for this gas?
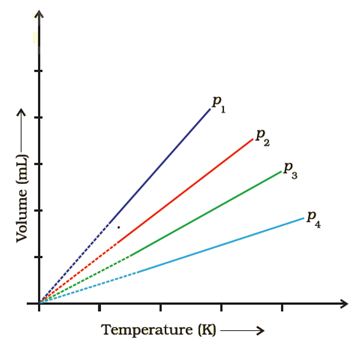

Answer : Option B
Explaination / Solution:
keep Temperature constant we know Pα1/V So as V increases P decreases.
Q4. Simple protein is.
Answer : Option B
Explaination / Solution:
Simple protein is Albumin.
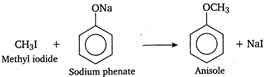
Q5. Anisole can be prepared by the action of methyl iodide on sodium phenate. The reaction is called
Answer : Option D
Explaination / Solution:
The
reaction of alkyi halide with sodium alkoxide to give ether (alkoxy
alkane), is known as Williamsons synthesis. In this reaction an ether
(anisole) is prepared by the action of alkyi halide (methyl iodide) on
sodium alkoxide (sodium phenate), so it is an example of Williamsons
synthesis.
Q6. The depression in freezing point for 1M urea, 1 M glucose and 1 M NaCl are in the ratio of
Answer : Option C
Explaination / Solution:
NaCl has i=2 while urea and glucose will have i=1
Q7. When one of the following reacts with NaOH, the product is sodium benzoate
Answer : Option B
Explaination / Solution:
Benzoic acid reacts with NaOH to form sodium benzoate.
Q8. Molecular formula represents
Answer : Option B
Explaination / Solution:
Molecular formula is the exact no. of atoms present in a molecule of a compound.
Molecular formula of a compound is related with its empirical formula as ,
Mollecular formula = ( Empirical formula ) n
where n represents a positive integer .
Q9. Presence of a nitro group in a benzene ring
Answer : Option B
Explaination / Solution:
This is because the Nitro is electron withdrawing group,it pulls the electron density from the ring towards itself thereby decreasing the electron density in the ring and deactivating the ring towards attack by the electrophile.
Q10. An ore has impurities which are lighter than the ore. The process used for the concentration of ore is
Answer : Option B
Explaination / Solution:
This is hydraulic washing or gravity separation. Here when stream of water is passed it takes away all the impurity with it
