Chemistry - Online Test
Q1.
For testing halogens in an organic compound with solution, sodium extract (Lassaigne’s test) is acidified with dilute What will happen if a student acidifies the extract with dilute in place of dilute HNO3?
Answer : Option D
Explaination / Solution:
white precipitate of is formed.
Q2. Activation energy of a reaction is
Answer : Option C
Explaination / Solution:
Activation energy is minimum energy required for reactant to get converted to product.
Q3. The number of phases in colloidal system are
Answer : Option A
Explaination / Solution:
Dispersed phase, dispersion medium
Q4. Which of the following property of water can be used to explain the spherical shape of rain droplets?
Answer : Option C
Explaination / Solution:
surface Tension is used.
Q5. Protein is.
Answer : Option D
Explaination / Solution:
Protein is condensation polymer.
Q6.
Mark the correct order of decreasing acid strength of the following compounds.
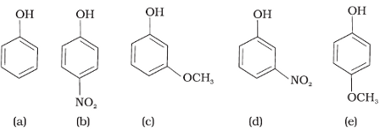
Answer : Option B
Explaination / Solution:
B will be most acidic because of –M effect of NO2 . Followed by d, in d –I effect of NO2 operates only. Then c will come as –I of OCH3 < - I of NO2 and least will be e because of +M effect of OCH3 that will decrease the acidity
Q7. 6.3 g oxalic acid is used to make 250 mL aqueous solution .The volume of 0.1N NaOH solution required to neutralize completely 10 mL of this solution is
Answer : Option C
Explaination / Solution:
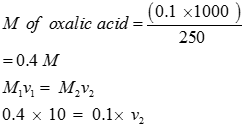
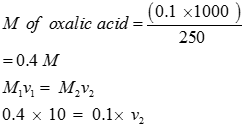
Q8. In a coupling reaction, the azo products obtained, involve an
Answer : Option D
Explaination / Solution:
In coupling reaction Diazonium salt to act as a electrophile and –N=N- bond forms.
Q9. SI unit of density is
Answer : Option D
Explaination / Solution:
Since, density
= mass / volume ;
and SI unit of mass is kg and that of volume is
using these SI units the unit of density is derived through dimensional analysis :
density = mass / volume
= kg / m3
or, = kg m-3
Q10. The reagent(s) for the following conversion, is/are
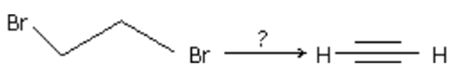
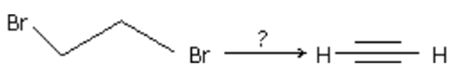
Answer : Option C
Explaination / Solution:
Alcoholic KOH followed by is used. Both are bases. is better base than alc.KOH.
