Chemistry - Online Test
Q1. Ferrous sulphate on heating gives
Answer : Option B
Explaination / Solution:
FeSO4 on heating gives SO2 and SO3
Q2. Reaction of carbon monoxide gas of syngas mixture with steam in the presence of catalyst is called
Answer : Option C
Explaination / Solution:
The water gas shift reaction converts carbon monoxide and water to carbon dioxide and hydrogen. The reaction is catalysed by a number of different base metal catalysts, depending on the operating temperature and levels of poisons in the feedstock.
Q3. Blue solution of CuSO4 becomes darker when treated with ammonia because
Answer : Option C
Explaination / Solution:
Ammonia forms stable complex with Cu2+
Q4. Narcotic drugs are used in medicinal doses because
Answer : Option C
Explaination / Solution:
Narcotic drugs are addictive.
Q5. The compound which forms acetaldehyde when heated with dilute NaOH is
Answer : Option D
Explaination / Solution:
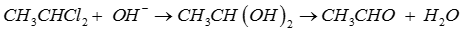
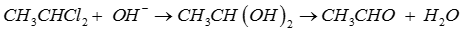
Q6.
For the reduction of silver ions with copper metal the standard cell potential was found to be +0.46V at 25°C. The value of standard Gibbs energy, ΔG° wll be (F = 96500 C )
Answer : Option A
Explaination / Solution:
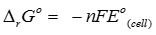
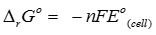
Q7. Thermodynamics is not concerned about
Answer : Option B
Explaination / Solution:
Thermodynamics on tells that whether reaction will give take place or not. It doesnot tell the rate at which reaction will occur.
Q8. The species having pyramidal shape is
Answer : Option B
Explaination / Solution:
SF2O has pyramidal shape as it is sp3 hybridised and it has 3bp and 1 lp.
Q9. A dibromo derivative of an alkane reacts with sodium metal to form an alicyclic hydrocarbon. The derivative is
Answer : Option D
Explaination / Solution:
This is intramolecular wurtz reaction.
Q10. Cement is an industrially important compound of
Answer : Option B
Explaination / Solution:
Cement is the important compound of Calcium.
