Chemistry - Online Test
Q1. Equanil is used to
Answer : Option A
Explaination / Solution:
It is used to relieve tension.
Q2. Which of the following pair contains complex salt and double salt respectively?
Answer : Option A
Explaination / Solution:
[Cu(NH3)4]SO4 this is complex salt while K2SO4Al2(SO4)3.24H2O is double salt.
Q3. Kohlrausch law of independent migration of ions states
Answer : Option A
Explaination / Solution:
Statement of Kohlrausch Law.
Q4. For , bond order is
Answer : Option C
Explaination / Solution:
Bond order is the number of chemical bonds between a pair of atoms. in diatomic nitrogen N≡N, Triple covalent bond is present in nitrogen so the bond order is 3;
Q5. Correct IUPAC name the following compound is
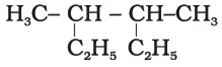
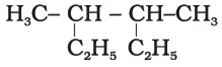
Answer : Option D
Explaination / Solution:
3,4- dimethylhexane will be correct iupac name as 6 membered carbon chain will be the main chain.
Q6. Molecular mass of a compound is the ------------- of atomic masses of elements present in a molecule .
Answer : Option B
Explaination / Solution:
Molecular mass is the sum of atomic masses of the elements present in a molecule.
For example ; Molecular mass of CO2 = atomic mass of one carbon atom & atomic mass of two oxygen atoms
=[ 12+2(16) ] u
= 44u.
Q7.
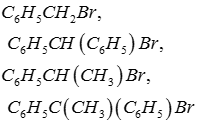
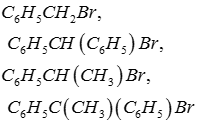
Answer : Option D
Explaination / Solution:
For SN1 stability of carbocation which matters. In C6H5C(CH3)(C6H5)Br carbocation will be tertiary and will be resonance stablised.
Q8. The minimum amount of energy required by the reacting molecules at the time of collisions in order to produce effective collisions is called
Answer : Option B
Explaination / Solution:
The minimum amount of energy required by the reacting molecules at the time of collisions in order to produce effective collisions is called threshold energy.
Q9. Substances that alter the rate of a chemical reaction without being used up in a chemical reaction is known as
Answer : Option D
Explaination / Solution:
Catalyst only increases or decreases the activation energy of a reaction. It doesnot get used up in reaction.
Q10. Increase in kinetic energy can overcome intermolecular forces of attraction. How will the viscosity of liquid be affected by the increase in temperature?
Answer : Option D
Explaination / Solution:
Viscosity is inversely proportional to temperature., hence viscosity of the given liquid decreases with rise in temperature since, the liquid particles can move more freely as the friction between them become lesser.
