Chemistry - Online Test
Q1. n the Wurtz reaction method of preparation of alkanes, the stoichiometric coefficient of Sodium is
Answer : Option A
Explaination / Solution:
Wurtz reaction is used for the preparation of alkanes from alkyl halides in the presence of sodium and dry ether.For example: CH3Br + 2Na +BrCH3 ---------> CH3CH3 + 2NaBr
Thus two atoms of Na are used.
Q2. Molarity of the solution is
Answer : Option B
Explaination / Solution:
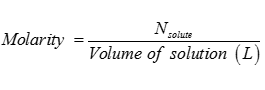
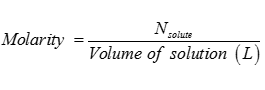
Q3. Redox reaction between copper and aqueous solution of silver nitrate happens when copper rod is dipped in silver nitrate solution. What happens in the final stage?
Answer : Option A
Explaination / Solution:
This is due to the formation of Cu2+ ions in the solution illustrated in following ionic reaction:
Cu(S)+2Ag+(aq)--------------˃Cu2+(aq)+2Ag(s)
Here, Cu is oxidized to Cu2+ (means relasing of 2 electrons from Cu to Cu2+ ) and Ag+ is reduced to Ag(s) (means gaining of 2 electrons from 2Ag+ to 2Ag ) and thus, silver converted into solid state and deposited on copper rod.
Q4. Example of ferromagnetic substance is
Answer : Option C
Explaination / Solution:
This is a Fact
Q5. Coordination number of hcp and ccp is
Answer : Option A
Explaination / Solution:
In hcp and ccp lattice, each atom is surrounded by 12 other atoms . Therefore, hcp and ccp lattice have ccordination no. 12.
Q6. It is now recognized that the ‘Modern Periodic Law’ is essentially the consequence of
Answer : Option A
Explaination / Solution:
Modern Periodic Law of element may be defined as the Physical and chemical properties of the elements are periodic functions of their atomic numbers.It means, when the elements are arranged in the order of their increasing atomic numbers, it is observed that the elements of similar properties recur at regular intervals or periodically. As a result of this, the elements fall in certain groups and lead to an arrangement called the modern periodic table of elements. It must be noted that elements are arranged in the periodic table in order of atomic numbers because atomic number is the most fundamental property of an element. The systematic arrangement of elements in modern periodic table according their atomic number helps in justification of isotopes of elements at one place.
Q7. Repeated addition of monomer molecule possessing double or triple bonds, in this case, propene, forms
Answer : Option C
Explaination / Solution:
This will form Polypropene.
Q8. In laboratory ammonia is prepared by heating
Answer : Option D
Explaination / Solution:
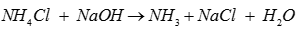
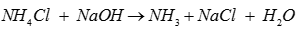
Q9. For isothermal reversible free expansion of an ideal gas from the initial volume to the final volume the work done on the gas is
Answer : Option D
Explaination / Solution:
this is workdone by the system in iosothermal process.
Q10. Which metal is also found in sea beds ?
Answer : Option C
Explaination / Solution:
Manganese is found in sea beds.
