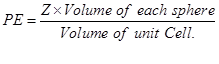Chemistry - Online Test
Q1. Alcoholic compounds react
Answer : Option D
Explaination / Solution:
The functional group of the alcohols is the hydroxyl group, –OH. Unlike the alkyl halides, this group has two reactive covalent bonds, the C–O bond and the O–H bond. The electronegativity of oxygen is substantially greater than that of carbon and hydrogen. Consequently, the covalent bonds of this functional group are polarized so that oxygen is electron rich and both carbon and hydrogen are electrophilic.
Q2. The tanks used by divers are filled with air diluted with
Answer : Option B
Explaination / Solution:
Size of Helium is small so does not causes “Bents” to divers when the dive back to surface.
Q3.
Name the following amine as per the IUPAC system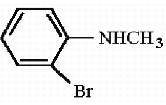
Answer : Option B
Explaination / Solution:
This is 2-bromo-N-methylaniline.
Q4.
Answer : Option D
Explaination / Solution:
sodium is oxidized and hydrogen is reduced.
Q5. The gram molar mass of is
Answer : Option C
Explaination / Solution:
Molar mass of CO2 = (atomic mass of C , 2*atomic mass of O )
= [12+2(16)] u
Since , gram molar mass
= Molar mass expressed in gms .
gram molar mass of CO2
= 44 g
Q6. Which of the following is thermally the most stable?
Answer : Option D
Explaination / Solution:
Stability of hydrides decreases down the group so most stable is H2O
Q7. The correct IUPAC name of the following alkane is
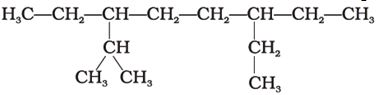
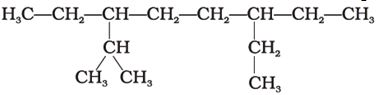
Answer : Option A
Explaination / Solution:
Following the rules of nomenclature,the IUPAC name of the given compound is 3,6-Diethyl-2-methyloctane
Q8. The Wilfley table is used in the process of
Answer : Option A
Explaination / Solution:
Wilfley table is used in Hydraulic washing.
Q9. The enthalpy of combustion of methane, graphite and dihydrogen at 298 K are, - 890.3 kJ mol-1, -393.5 kJ mol-1 and –285.8 kJ mol-1 respectively. Enthalpy of formation of CH4 (g) will be
Answer : Option C
Explaination / Solution:
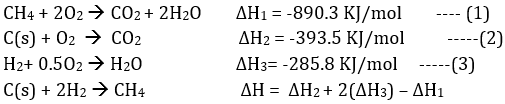
ΔH= -393.5 +2(-285.8)-(-890.3)
= -74.8 kJ/mo
Q10. Packing efficiency in body centered cubic structure is
Answer : Option B
Explaination / Solution: