Chemistry - Online Test
Q1. Mohr’s salt is a
Answer : Option D
Explaination / Solution:
Mohr salt is an example of double salt. Mohr Salt is FeSO4. (NH4)2SO4.6H2O.
Q2. The aldehydes and ketones undergo one of the following reactions.
Answer : Option B
Explaination / Solution:
Aldehydes and Ketones undergoes nucleophilic addition reaction .
Q3. Molarity is defined as,
Answer : Option C
Explaination / Solution:
Molarity is the most widely used method of expressing the strength or concentration of a solution .
It is denoted by ' M ' .
Molarity is defined as number of . of moles of solute present per litre of solution.
Mathematically ,
Molarity ( M )
= Number of moles of solute / Volume of solution in Litres
Q4. Presence of extensive hydrogen bonding between water molecules is responsible for
Answer : Option A
Explaination / Solution:
hydrogen bonding is responsible for liquid and solid states.
Q5. Chlordiazepoxide, meprobamate and equanil are
Answer : Option D
Explaination / Solution:
Theya re tranquillisers.
Q6. In which of the following complexes, the nickel metal is in the highest oxidation state?
Answer : Option C
Explaination / Solution:
In A Ni has +4 oxidation state.
Q7. Fuel cells are ________ that are designed to convert the energy of combustion of fuels like hydrogen, methane, methanol, etc. directly into_________.
Answer : Option A
Explaination / Solution:
Fuel Cells converts energy coming from the combustion of fuels directly to electrical energy.
Q8. In NO-3 ion, the number of bond pairs and lone pairs of electrons on nitrogen atom are
Answer : Option C
Explaination / Solution:
The nitrate ion is formed by the loss of the hydrogen ion, and so its structure is:
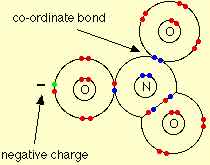
Around the central nitrogen there are 4 pairs of shared electrons, and no remaining lone pair. The original lone pair has now become a bonding pair. Two of those pairs make up a double bond. The double bond unit and the two single bonds arrange themselves as far apart as possible in a trigonal planar arrangement - exactly the same as the carbonate ion.
Q9. In the reaction
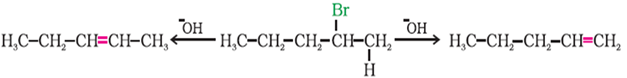
Pent – 2 – ene. 2 – Bromopentane. Pent – 1 – ene, 2 – Bromopentane on heating with alcoholic KOH, forms two compounds: Pent – 1 – ene and Pent – 2 – ene., if one major and one product are formed , then
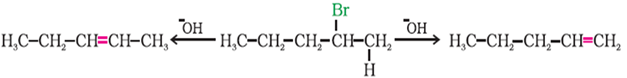
Pent – 2 – ene. 2 – Bromopentane. Pent – 1 – ene, 2 – Bromopentane on heating with alcoholic KOH, forms two compounds: Pent – 1 – ene and Pent – 2 – ene., if one major and one product are formed , then
Answer : Option B
Explaination / Solution:
The major alkene will ne more substituted that what zaitsev rule is.
Q10. What is the type of hybridisation of each carbon in the compound
Answer : Option B
Explaination / Solution:
C in CH3 is and C of C=O bond is .
