Chemistry - Online Test
Lewis developed electronic theory of valence or theory of chemical bonding to explain the formation of chemical bond between the two atoms.
According to the electronic theory of valence, every atom tries to attain octet configuration (presence of eight electrons) in its valence shell by losing or gaining or by sharing of electrons. This is known as the "Octet Rule".
The electrostatic forces of attraction that holds the two oppositely charged ions together are known as "electrovalent bond".
in hybridization as % of 's'character increases electronegativity also increases.
more the s charater, more will be the electronegativity of that atom.
increasing order ofelectronegativity with %s character is sp (50%) > sp2(33.33%) > sp3(25%)
Oxidation means addition of oxygen, removal of hydrogen, increase in oxidation number, addition of a electronegative element or removal of an electropositive element
The term oxidation was first used to describe chemical reactions in which oxygen was added to an element of a compound. Later on the term oxidation was extended to describe many more reactions which occurred without the use of even oxygen.
Addition of Oxygen: Oxidation is a chemical reaction in which oxygen is added to any chemical species (atom, ion or molecule). Example
2Mg + O2 → 2MgO
S + O2 → SO2
2CO + O2 → 2CO2
Na2SO3 + H2O2 → Na2SO4 + H2O
Removal of Hydrogen : Oxidation is a chemical reaction in which hydrogen is removed from any chemical species (atom, ion or molecule).
H2S + Cl2 → 2HCl + S
4HI + O2 → 2H2O + 2I2
4HI + MnO2 → MnCl2 + 2H2O + Cl2
Addition of an Electronegative Element: Oxidation is a chemical reaction in which an electronegative element is added into any chemical species (atom, ion or molecule).
Fe+ S → FeS (Oxidation of iron)
SnCl2 + Cl2→ SnCl4(Oxidation of stannous chloride)
2Fe + 3F2 → 2FeF3(Oxidation of iron)
Removal of an Electropositive Element: Oxidation is a chemical reaction in which an electropositive element is removed from any chemical species (atom, ion or molecule).
2KI + H2O2 → 2KOH + I2 (Oxidation of potassium iodide)
2K2MnO4 + Cl2 → 2KCl + 2KMnO4 (Oxidation of potassium manganate)
2KI + Cl2 → 2KCl + I2 (Oxidation of potassium iodide)
A substance which brings oxidation is known as oxidizing agentt
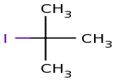 actually
here firstly we select longest chain and it is of 3 carbon.and at
number 2 position there is iodo as well as methyl ,so its name is 2-iodo
2-methyl propane.
actually
here firstly we select longest chain and it is of 3 carbon.and at
number 2 position there is iodo as well as methyl ,so its name is 2-iodo
2-methyl propane.is 2 – Iodo 2·methyl propane
