Chemistry - Online Test
Q1. The crystals of ferrous sulphate on heating give
Answer : Option D
Explaination / Solution:
FeSO4 on heating gives Fe2O3+SO2+SO3+H2O.
Q2. The polymer with the amide linkage is
Answer : Option D
Explaination / Solution:
Nylon has amide linkage.
Q3. The decomposition of hydrogen peroxide to form water and oxygen is an example of
Answer : Option C
Explaination / Solution:
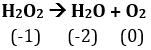
Oxidation number of O in is -1 and it is decreasing to -2 to and increases to 0 in
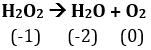
Oxidation number of O in is -1 and it is decreasing to -2 to and increases to 0 in
Q4. MgO has the structure of NaCl. The coordination number of the ions in MgO is
Answer : Option D
Explaination / Solution:
Coordination No. in NaCl is 6.
Q5. Which of the following is not an actinoid?
Answer : Option D
Explaination / Solution:
Terbium (Z=65) has an electronic configuration of [Xe] 4f9 6s2. It is a member of 4f-inner transition series which is called lanthanoid series. Rest option are all the members of 5f-inner transition series, also known as the actinoid series i.e. Curium ([Rn] 5f7 6d1 7s2) ; Californium ([Rn] 5f10 7s2); and Uranium ([Rn] 5f3 6d1 7s2).
Q6. Photochemical smog occurs in warm, dry and sunny climate. One of the following is not amongst the components of photochemical smog, identify it.
Answer : Option D
Explaination / Solution:
is not the component of photochemical smog.
Q7. Values of e/m (charge/mass) in the categories alpha particle (α), electron (e) and protons (p) increase in the order:
Answer : Option B
Explaination / Solution:
-particle (He2+) has very high mass compared to proton and electron, therefore very small ratio. Proton and electron have same charge (magnitude) but former is heavier, hence has smaller value of .
Q8. The compound which is widely used as a white pigment is
Answer : Option D
Explaination / Solution:
ZnO is white in color.
Q9. Only one element of ________ forms hydride.
Answer : Option C
Explaination / Solution:
Only Chromium (Cr) who belongs to group 6 forms hydride.
Elements of group 7, 8, 9 of d – block do not form hydrides at all. This inability of metal, of group 7, 8, 9 of periodic table to form hydrides is referred to as hydride gap of d – block.
Q10. The IUPAC name of KAl(SO4)2.12H2O is
Answer : Option C
Explaination / Solution:
This is double salt so name is written in sequential manner.
