Chemistry - Online Test
Q1.
In the reaction,
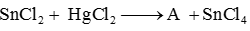 A is
A is
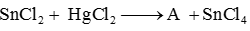 A is
A is
Answer : Option B
Explaination / Solution:
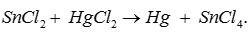
Q2. Self-ionization of water is called
Answer : Option A
Explaination / Solution:
In auto-protolysis a proton is transferred between two identical molecules, one of which acts as a Bronsted acid, releasing a proton which is accepted by the other molecule acting as a Bronsted base.
Auto-protolysis is also called the self-ionization. Auto-protolysis of water is a reaction in which self-ionization of water molecules takes place to produce hydronium ion (H3O+) and hydroxide ion (OH-).
Q3. According to Werner’s theory, the secondary valences of the central atom correspond to its
Answer : Option D
Explaination / Solution:
According to Werner’s theory CN is equal to secondary valency.
Q4. For making distinction between 2 – pentanone and 3 – pentanone the reagent to be employed is
Answer : Option D
Explaination / Solution:
2-pentanone will give iodoform but 3-pentanone will not give.
Q5. Potassium metabisulphite is a food – preservative as it is
Answer : Option D
Explaination / Solution:
produces
Q6. Which of the following electrolytic solutions has the least specific conductance?
Answer : Option D
Explaination / Solution:
Specific conductance decreases with dilution because of decrease in the number of ions per unit volume.
Q7. Which of the following statements is correct?
Answer : Option D
Explaination / Solution:
It will be a closed system.
Q8. For a stable molecule the value of bond order must be
Answer : Option B
Explaination / Solution:
Bond order∝Stability
Hence, for a stable molecule the value of bond order must be positive. When bond order is zero and negative the molecule will not exist.
Q9. The synthesis of 3 – octyne is achieved by adding a bromoalkane into a mixture of sodium amide and an alkyne. The bromoalkane and alkyne respectively are
Answer : Option D
Explaination / Solution:
NaNH2 is very strong base it will abstract H from alkyne and then substitution reaction will occur.
Q10. The alkali metals are low melting. Which of the following alkali metal is expected? to melt if the room temperature rises to
Answer : Option C
Explaination / Solution:
Cs has very low melting point ()
