Chemistry - Online Test
Q1. The addition of HCl to an alkene proceeds in two steps. The first step is the attack of H+ ion to portion which can be shown as
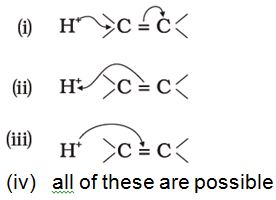
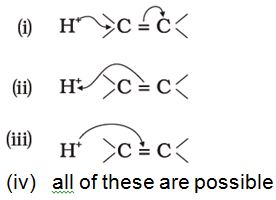
Answer : Option C
Explaination / Solution:
arrows are shown in the direction of movement of electrons.
Q2. The substance that slows down the reaction without being consumed is known as
Answer : Option B
Explaination / Solution:
Inhibitors inhibits the action of catalyst and hence slows down the rate of reaction.
Q3. A biochemical catalyst is
Answer : Option C
Explaination / Solution:
Enzymes are biological catalyst, they catalize different chemical reactions in body.
Q4. A person living in Shimla observed that cooking food without using pressure cooker takes more time. The reason for this observation is that at high altitude:
Answer : Option C
Explaination / Solution:
As we move to higher altitude P decreases.
Q5. The carrier of hereditary character is.
Answer : Option D
Explaination / Solution:
Nucleotides is carrier of hereditary
Q6. Building unit of protein is.
Answer : Option A
Explaination / Solution:
Protein is built by α – Aminoacid
Q7. Which of the following is most acidic?
Answer : Option C
Explaination / Solution:
In cases of halogen derivatives of phenols or aniline or benzoic acid etc, it is very helpful to understand that all halogens when attached to benzene ring exerts -I as well as +R effect.
In case of Cl, Br and I, the +R effect has almost no effect on reactivity, acidic character or basic character of benzene ring. It is due to very less effective overlapping involving 2p of carbon and 3p or 4p or 5p of halogen.
Hence, only -I effect becomes the deciding factor, which is most dominant from ortho-position and least effective from para-position. So m chlorophenol is most acidic.
Q8.
Calculate the molarity of liquid HCl solution whose density is 1.17 g/cm3
.
Answer : Option C
Explaination / Solution:
M= (1.17 x 1000)(36.5)
Q9. Anisole reacts with a mixture of concentrated sulphuric and nitric acids to yield a mixture of ortho and para nitroanisole
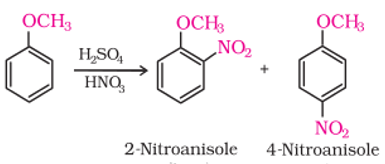

Answer : Option C
Explaination / Solution:
Para isomer is major because ethere is no steric hinderance and also at para +M effect will work effectively.
Q10. Chemistry does not play a major role in
Answer : Option A
Explaination / Solution:
Chemistry does not deal in explaining superconductivity.
*In superconducting materials the charecteristics of superconductivity appear when the temperature is lowered below a critical temperature.
*The onset of superconductivity is accompanied by abrupt changes in physical properties which are more related to phase transitions of the material .
These aspects of studies in properties of materials are better related to studies in the fields of Physics , eventhough principles involved in Chemistry & Physics go hand to hand.
