Chemistry - Online Test
Q1. For an isolated system, ΔU = 0, what will be ΔS?
Answer : Option A
Explaination / Solution:
for isolated system ΔS = 0.
Q2.
How much electricity is required in coulomb for the oxidation of 1 mol of FeO to ?
Answer : Option C
Explaination / Solution:
For converting FeO to 1mol of electrons are required.
Q3.
Hybridization of and of are
Answer : Option A
Explaination / Solution:
C2 is sp2 while C3 is sp (special case of allenes)
Q4. Chloroform is in closed dark coloured bottles completely filled
Answer : Option D
Explaination / Solution:
In presence of light choloroform oxidizes to form phosgene , which is poisonous gas.
Q5. Beryllium carbonate is unstable and can be kept only in the atmosphere of
Answer : Option C
Explaination / Solution:
is very unstable due to size incompatibility.
Q6.
Sodium cyanide, sulphide and halide,( -CN, -S and -H, coming from organic compound) so formed on sodium fusion are extracted from the fused mass by boiling it with distilled water. This is called
Answer : Option A
Explaination / Solution:
This is lassaignes test.
Q7. The rate constant of the reaction at temperature 200 K is 10 times less than the rate constant at 400 K. The activation energy of the reaction is
Answer : Option C
Explaination / Solution:
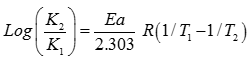
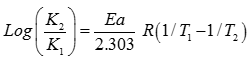
Q8. Hydrophilic colloids are stable due to
Answer : Option C
Explaination / Solution:
This makes the colloid stable.
Q9. The van der Waals Equation adjusts the measured pressure
Answer : Option B
Explaination / Solution:
/ is added.
Q10. Bees – wax is.
Answer : Option A
Explaination / Solution:
Bees wax is myricylpaimitate.
