Chemistry - Online Test
Q1. Temporary hardness It can be removed in boiling by precipitating
Answer : Option C
Explaination / Solution:
.Mg can be precipitate out in order to remove temporary hardness.
Q2. The appropriate reagent for the transformation:
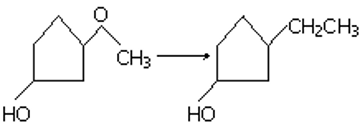

Answer : Option D
Explaination / Solution:
For this reaction wolff Kishner reaction is used as –OH is acid sensitive group so clemmson cannot be used.
Q3. Which of the following complex ions/molecules of nickel is paramagnetic?
Answer : Option A
Explaination / Solution:
Ni2+ is in 3d8 configuration so its para magnetic .
Q4. The purpose of adding bithional to medicated soaps is
Answer : Option C
Explaination / Solution:
bithional impart antiseptic properties.
Q5. How much electricity in terms of Faraday is required to produce 20.0 g of Ca from molten CaCl2?
Answer : Option B
Explaination / Solution:
20g is 0.5 mol of and for 1mol 2F charge is required.
Q6. The hybridization of orbitals of N atom in, and are respectively
Answer : Option C
Explaination / Solution:
Hybridization of orbitals of N atom in, and are
Q7. The most common freons in industrial use is manufactured by
Answer : Option D
Explaination / Solution:
Most common Freon is synthesized by Swartz reaction.
Q8. Alkaline earth metals have large size of the atoms. Therefore, they shows
Answer : Option C
Explaination / Solution:
The reason is that the atoms of alkali metals are of large sizes. therefore, the outermost electon is for away from the nucleus and canbe easily removed
Q9. The elements present in an organic compound are converted from covalent form into the ionic form by fusing the compound with sodium metal. This test is called
Answer : Option A
Explaination / Solution:
to covert covalent form to ionic form sodium fusion test is done this is lassaignes test.
Q10. Which among the following is an example of photochemistry used in our daily life?
Answer : Option B
Explaination / Solution:
Photography is an example of photochemistry used in our daily life. Reaction takes place in the presence of light
