General Science - Online Test
Q1. the valence shell electron pair repulsion (vsepr) theory helps in the
Answer : Option A
Explaination / Solution:
Valence shell electron pair repulsion (VSEPR) theory is a model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. It is also named the Gillespie-Nyholm theory after its two main developers.
Q2. There are two stationary coils near each other. If current in coil-1 is changing with time, relationship of current in coil-2 to it's emf is described by
Answer : Option D
Explaination / Solution:
M is mutual inductance
Q3. Davisson Germer experiment proves that
Answer : Option B
Explaination / Solution:
No Explaination.
Q4. Correct sequence for the amount of dissolved oxygen in water body is:
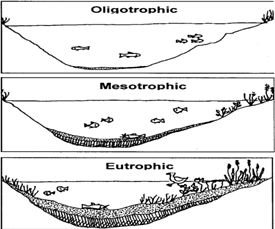

Answer : Option A
Explaination / Solution:
Dissolve oxygen in water bodies is utilized by aquatic organisms for respiration. Oligotrophic have least dissolved oxygen followed by mesotrophic and eutropic have highest concentration of dissolved oxygen.
Q5. A mixture containing two enantiomers in equal proportions
Answer : Option D
Explaination / Solution:
For a mixture of two enantiomers , one rotates the plane polarized light towards left and other towards right therefore there is no optical rotation and hence mixture is optically inactive.
Q6.
The molecular formula represents two alcohols: propan-1-ol and propan-2-ol. This property is called as:
Answer : Option D
Explaination / Solution:
propan-1-ol and propan-2-ol are positional isomers as they differ only in the position of functional group -OH.
Positional isomers are isomers that have the same carbon skeleton and the same functional groups but differ from each other in the location of the functional groups on or in the carbon chain.
Q7. The basic operations performed by a com-puter are
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. The alkali metals burn vigorously in oxygen forming oxides. Hence, potassium forms
Answer : Option A
Explaination / Solution:
alkali metals burns vigorously to form superoxides. Superoxide O2- ion is only in the presence of large cations such as potassium, Rb and Cs
Q9. Attenuation is
Answer : Option D
Explaination / Solution:
Attenuation is a general term that refers to any reduction in the strength of a signal. Attenuation occurs with any type of signal, whether digital or analog. Sometimes calledloss, attenuation is a natural consequence of signal transmission over long distances.
Q10. APS= 1+APC. It is
Answer : Option C
Explaination / Solution:
No Explaination.
