General Science - Online Test
Q1. The term reduction has one of the following options taking place.
Answer : Option D
Explaination / Solution:
Reduction is addition of hydrogen, removal of oxygen, removal of an electronegative element or addition of an electropositive element.
- Removal of Oxygen: Reduction is a chemical reaction in which oxygen is removed from any chemical species (atom, ion or molecule).
CuO + C → Cu + CO
H2O + C → CO + H2
2CO + O2 → 2CO2
Fe3O4 + 4H2 → 3F2 + 4H2O - Addition of Hydrogen : Reduction is a chemical reaction in which hydrogen is added to any chemical species (atom, ion or molecule).
Cl2 + H2 → 2HCl
S + H2 → H2S
C2H4 + H2 → C2H6 - Removal of an Electronegative Element: Reduction is a chemical reaction in which an electronegative element is removed from any chemical species (atom, ion or molecule).
2HgCl2 + SnCl2 → Hg2Cl2 + SnCl4 (Reduction of mercuric chloride)
2FeCl3 + H2 → 2FeCl2 + 2HCl (Reduction of ferric chloride)
2FeCl3 + H2S → 2FeCl2 + 2HCl + S (Reduction of ferric chloride)
- Addition of an Electropositive Element: Reduction is a chemical reaction in which an electropositive element is added any chemical species (atom, ion or molecule).
HgCl2 + Hg → Hg2Cl2 (Reduction of mercuric chloride)
CuCl2 + Cu → Cu2Cl2 (Reduction of cupric chloride) - The substance which brings reduction is known as reducing agent.
- A substance, which undergoes oxidation, acts as a reducing agent while a substance, which undergoes reduction, acts as an oxidizing agent.
Mg, S, Cu, Na2SO3, H2S, HI, H2, C, KI are reducing agents, while O2, Cl2, F2, H2O2, MnO2, FeCl3, CuCl2, Fe3O4, CuO, etc., are oxidizing agents in the above examples.
- All oxidation and reduction reactions are complimentary of one another and occur simultaneously, one cannot take place without the other.
- No single oxidation and no single reduction process is known. The simultaneously oxidation and reduction reactions are generally termed as redox reactions. e.g., 2FeCl3 + SnCl2 → 2FeCl2 + SnCl4.
In above example iron undergoes reduction from +3 to +2.
Q2. Group I elements of the periodic table are collectively known as
Answer : Option D
Explaination / Solution:
Group I ( )elemens react vigorously with water to produce an alkaline solution thus colectively known as alkali metals
Q3. Ethanol is commercially produced through a particular species of
Answer : Option A
Explaination / Solution:
Saccharomyces is an yeast which is used to ferment sugar to produce ethanol in absence of oxygen or by anaerobic respiration.
Q4. In pure translational motion of a rigid body
Answer : Option C
Explaination / Solution:
In translational motion when the body moves along a straight line or more exactly when every point of the body travels on paralell lines, thus at any instant of time every particle of the body has the same velocity.
Q5. When a bar magnet is pushed towards the coil connected in series with a galvanometer, the pointer in the galvanometer G
Answer : Option B
Explaination / Solution:
When bar is in motion then flux will changed hence current will be induced.
Q6. In which case is electron emission from a metal not known?
Answer : Option A
Explaination / Solution:
No Explaination.
Q7. The word Science originates from the Latin verb Scientia meaning
Answer : Option B
Explaination / Solution:
The phrase "scientia potentia est" (or "scientia est potentia" or also "scientia potestas est") is a Latin aphorism meaning "knowledge is power"
Q8. A sure indicator of air pollution is
Answer : Option A
Explaination / Solution:
Bioindicators are living organisms that respond in an especially clear way to a change in the environment.
The lichens are useful bioindicators for air pollution, espeially sulfur dioxide pollution, since they derive their water and essential nutrients mainly from the atmosphere rather than from the soil.
The polluted air have very less or no lichens.
Q9. Give IUPAC names of the following compound:
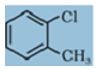

Answer : Option C
Explaination / Solution:
Chloro is given priority so umbered as 1
Q10. IBM 1401 is the first computer to enter in Nepal. It belonged to
Answer : Option B
Explaination / Solution:
No Explaination.
