Chemistry - Online Test
Q1. Which one of the following structures represents nylon 6,6 polymer?
Answer : Option D
Explaination / Solution:
No Explaination.
Q2. Conjugate
base for Bronsted acids H2O and HF are
Answer : Option C
Explaination / Solution:
H2O + H2O ↔ H3O+
+ OH-
acid 1 + base 1 ↔ acid 2 +
base 2
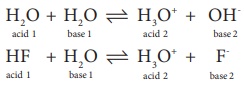
HF + H2O ↔ H3O+
+ F-
acid 1 + base 1 ↔ acid 2 +
base 2
Conjugate
bases are OH- and F- respectively
Q3. Which of the metal is extracted by Hall-Heroult process?
Answer : Option A
Explaination / Solution:
No Explaination.
Q4. 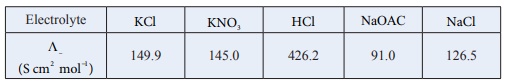

Calculate
ΛºHOAC using appropriate molar
conductances of the electrolytes listed above at infinite dilution in water at
25oC .
Answer : Option C
Explaination / Solution:
( Λº )HoAC = ( Λº)HCl + ( Λº)NaOAC - ( Λº)NaCl
= (426.2 + 91) −(126.5)
= 390.7
Q5. In diborane, the number of electrons that accounts for banana bonds is
Answer : Option C
Explaination / Solution:
There are two 3c –
2e-bonding in the bridges account for 4 electrons.
Q6. Statement : To stop bleeding from an injury, ferric chloride can be applied. Which comment about the statement is justified?
Answer : Option B
Explaination / Solution:
No Explaination.
Q7. On hydrolysis, PCl3
gives
Answer : Option A
Explaination / Solution:
No Explaination.
Q8. 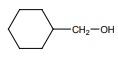 on treatment with Con H2 SO4 , predominately gives
on treatment with Con H2 SO4 , predominately gives
 on treatment with Con H2 SO4 , predominately gives
on treatment with Con H2 SO4 , predominately gives
Answer : Option B
Explaination / Solution:
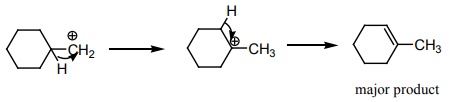

Q9. Which of the following compounds is colourless?
Answer : Option B
Explaination / Solution:
Ti4+ contains no
unpaired electrons in d orbital, hence no d-d transition.
Q10. 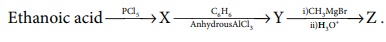
Predict the product Z in the following series of reactions

Answer : Option A
Explaination / Solution:


