Chemistry - Online Test
Q1.
Assertion : Coagulation power of Al3+ is more than Na+
.
Answer : Option A
Explaination / Solution:
(Hardy-Schulze rule)
Q2. In the brown ring test, brown colour of the ring is due to
Answer : Option B
Explaination / Solution:
No Explaination.
Q3. Which one of the following is the strongest acid
Answer : Option C
Explaination / Solution:
No Explaination.
Q4. The magnetic moment of Mn2+
ion is
Answer : Option A
Explaination / Solution:
Mn2+ => 3d5
contains 5 unpaired electrons
n=5 ; … = √n(n+2) BM
= √[5(5+2)] = √35 = BM
Q5. 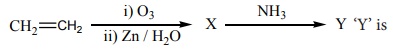
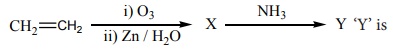
Answer : Option C
Explaination / Solution:
X-HCHO
Y-(CH2 )6N4
Q6. As per IUPAC guidelines,
the name of the complex [Co( en)2 ( ONO)Cl ] Cl is
Answer : Option D
Explaination / Solution:
No Explaination.
Q7. 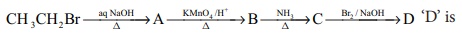
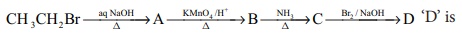
Answer : Option C
Explaination / Solution:
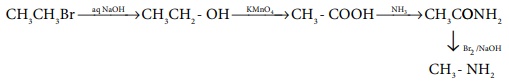
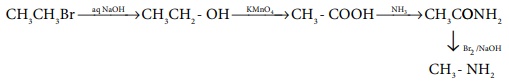
Q8.
Assertion : monoclinic
sulphur is an example of monoclinic crystal system
Answer : Option A
Explaination / Solution:
No Explaination.
Q9.
Assertion: A solution of sucrose in water is
dextrorotatory. But on hydrolysis in the presence of little hydrochloric acid,
it becomes levorotatory.
Reason: Sucrose hydrolysis gives equal amounts of glucose and fructose.
As a result of this change in sign of rotation is observed.
Answer : Option A
Explaination / Solution:
No Explaination.
Q10.
For the reaction, 2NH3 → N2 + 3H2
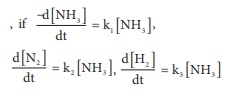
Answer : Option C
Explaination / Solution:
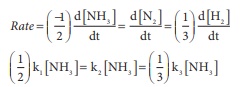
(3/2)k1 = 3k2
= k3
1.5 k1 = 3k2 = k3
