Chemistry - Online Test
x/m =k.p1/n
log(x/m )=logk+ 1/ n logp
y=c+mx
m = 1/n and c= logk
2 R - OH + 2Na→ 2 RONa + H2↑2 moles of alcohol gives 1 mole of H2
which occupies 22.4L at 273K and 1 atm
∴ number of moles of alcohol = ( 2
moles of R - OH / 22.4 L of H2 ) × 560 mL
= 0.05 moles
∴ no. of moles = mass / molar mass
⇒ molar mass = 3.7/0.05 = 74 g mol−1
General
formula for R - OH Cn H2n+1 - OH
∴ n(12) + (2n+1) (1) + 16+1=74
14n = 74 – 18
14n = 56
∴ n = 56/14 = 4
The 2 alcohol which contains 4
carbon is CH2 CH(OH)CH2CH3
 is
isIn the complex [M (en)2 (Ox)]
Cl For the central metal ion M3+
The primary valance is = +3
The secondary valance = 6
sum of primary valance and secondary valance = 3+6 = 9
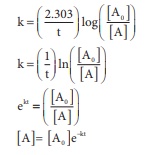
In this case
k = x min−1 and [A0]
= 0.01M = 1 × 10−2M
t = 1 hour = 60 min
[A]= 1 × 10−2 ( e−60x)
