Chemistry - Online Test
Q1. Glucose is an aldose. Which one of the following reactions is not expected with glucose?
Answer : Option B
Explaination / Solution:
No Explaination.
Q2. If the initial concentration of the reactant is doubled, the time for half reaction is also doubled. Then the order of the reaction is
Answer : Option A
Explaination / Solution:
For a first order reaction
t1/2 is independent of initial concentration .i.e., ∴ n ≠ 1; for such cases
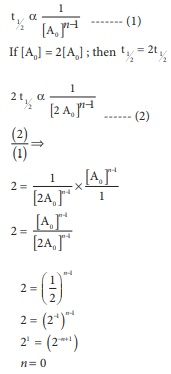
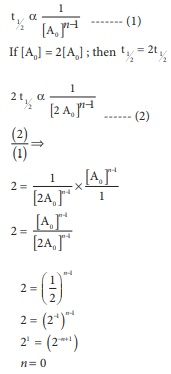
Q3. The pH of an aqueous solution is Zero. The solution is
Answer : Option B
Explaination / Solution:
pH = -log10[H+
]
∴[H+ ]=10-pH
=100
=1
[H+] = 1M
The solution is strongly acidic
Q4. The incorrect statement among the following is
Answer : Option D
Explaination / Solution:
No Explaination.
Q5. In
the electrochemical cell: Zn | ZnSO4 (0.01M) || CuSO4
(1.0M) | Cu , the emf of this Daniel cell is E1. When the
concentration of is changed to 1.0M and that CuSO4 changed to 0.01M,
the emf changes to E2. From the above, which one is the relationship
between E1 and E2?
Answer : Option B
Explaination / Solution:
Zn(s) → Zn2+ (aq) + 2e-
Cu2+ (aq)+2e-
→ Cu(s)
Zn(s) +Cu2+ (aq) → Zn2+
(aq) + Cu(s)
Ecell =Eºcell – (0.0591/2) log ( [zn2+]/
[Cu2+] )
E1 =Eºcell - 0.0591/2 log (10-2/1)
E1 =Eocell
+ 0.0591 ........(1)
E2 =Eºcell - 0.0591/2 log (1/10-2)
E2 =Eocell
- 0.0591 .........(2)
E1>E2
Q6. Williamson synthesis of preparing dimethyl ether is a / an /
Answer : Option B
Explaination / Solution:
No Explaination.
Q7. Which of the following oxidation states is most common among the lanthanoids?
Answer : Option D
Explaination / Solution:
No Explaination.
Q8. Phenyl methanal is reacted with concentrated NaOH to give two products X and Y. X reacts with metallic sodium to liberate hydrogen X and Y are
Answer : Option C
Explaination / Solution:
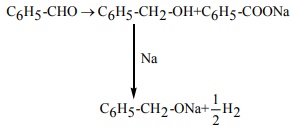

Q9. 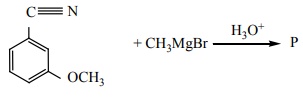 Product
‘P’ in the above reaction is
Product
‘P’ in the above reaction is
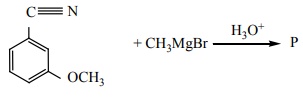 Product
‘P’ in the above reaction is
Product
‘P’ in the above reaction is
Answer : Option B
Explaination / Solution:
No Explaination.
Q10. Schottky defect in a crystal is observed when
Answer : Option B
Explaination / Solution:
No Explaination.
