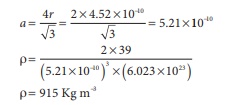Chemistry - Online Test
Q1. Conductivity
of a saturated solution of a sparingly soluble salt AB (1:1 electrolyte) at
298K is 1 .85 ×10−5 S m−1 . Solubility product of
the salt AB at 298K ( Λºm )AB = 14 × 10−3 S m2 mol−1 .
Answer : Option D
Explaination / Solution:
No Explaination.
Q2. The stability of +1 oxidation state increases in the sequence
Answer : Option A
Explaination / Solution:
stability of +1 oxidation state decreases down the group due to inert pair effect
Q3.
Match the following
A) Pure nitrogen i) Chlorine
B) Haber process ii) Sulphuric acid
C) Contact process iii) Ammonia
D) Deacons Process iv) sodium azide (or) Barium azide
Answer : Option D
Explaination / Solution:
No Explaination.
Q4. When copper is heated with
conc HNO3 it produces
Answer : Option C
Explaination / Solution:
No Explaination.
Q5. Among the following ethers which one will produce methyl alcohol on treatment with hot HI?
Answer : Option A
Explaination / Solution:
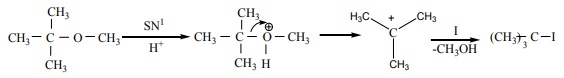
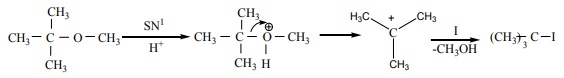
Q6. Which of the following lanthanoid ions is diamagnetic?
Answer : Option B
Explaination / Solution:
Yb2+ - 4f14
–no unpaired electrons – diamagnetic
Q7. The reagent used to distinguish between acetaldehyde and benzaldehyde is
Answer : Option B
Explaination / Solution:
No Explaination.
Q8. Choose the correct statement.
Answer : Option D
Explaination / Solution:
V2+ ( t2g3, eg0 ; CFSE =
3 x (-0.4)Δ0 = -1.2 Δ0)
Ti2+ ( t2g2, eg0 ; CFSE =
2 x (-0.4)Δ0 = -0.8 Δ0)
Statements given in
option (a) ,(b), and (c) are wrong.
The current statements
are
(a) since, the crystal
field stabilization is more in octahedral field , octahedral complexes are more
stable than square planar complexes.
(b) 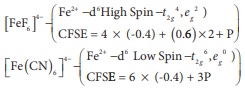
Q9. IUPAC
name for the amine
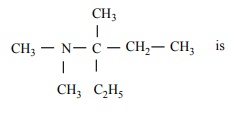
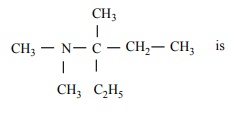
Answer : Option D
Explaination / Solution:
No Explaination.
Q10. Potassium has a bcc structure with nearest neighbor distance 4.52 Aº . its atomic weight is 39. its density will be
Answer : Option A
Explaination / Solution:
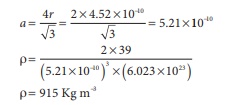
ρ = n × M / a3NA
for bcc
n = 2
M=39
nearest distance 2r =
4.52